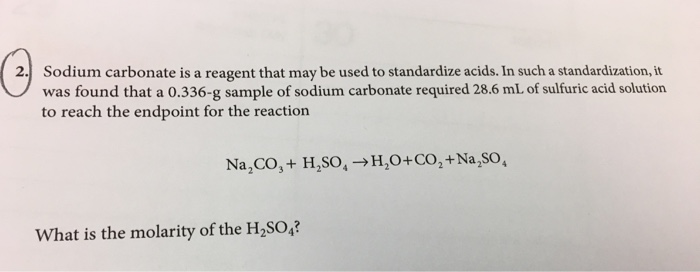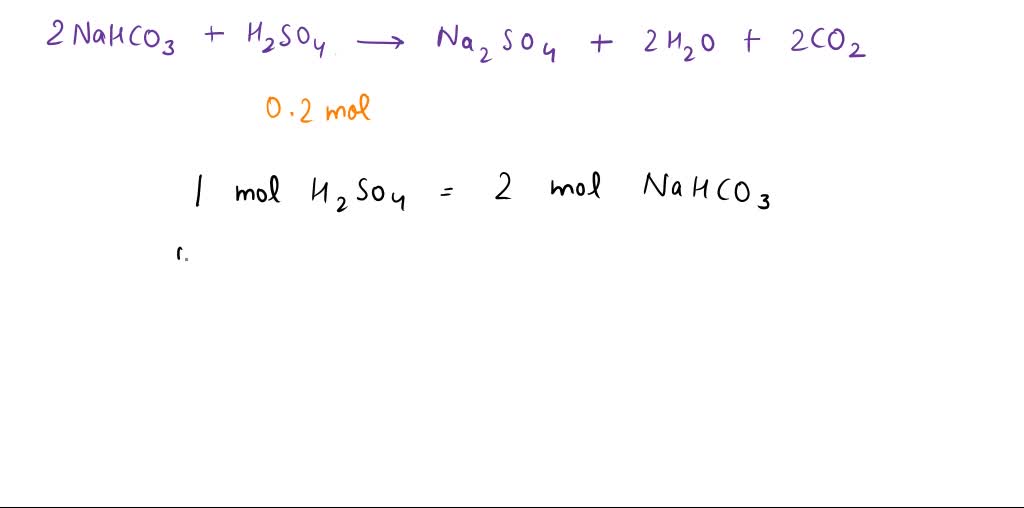
SOLVED: Sodium hydrogen carbonate (NaHCO3) reacts with sulfuric acid (H2SO4) to form sodium sulfate, carbon dioxide and water. What is the mass of sodium hydrogen carbonate required to neutralize 0.200 moles of
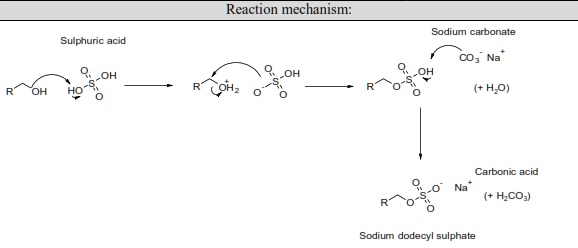
SOLVED: Reaction mechanism: Sodium carbonate Sulphurc acid CO3" Na COHz (+HJO) Carbonic acid HCO;) Sodium dodecyl sulphate
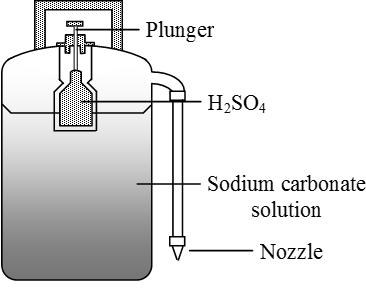
36. Assertion : Sodium Carbonate can be titrated against sulphuric acid by using methyl orange as indicator. Reason : The Volume of Sulphuric Acid required to produce the Colour change for the
If 25ml of a sulphuric acid solution reacts completely with 1.06 g of pure sodium carbonate, what is the normality of this acid solution?

H 2 SO 4 + Zn 1) Sulphuric acid + zinc 3) Nitric acid + sodium thiosulphate 2) Hydrochloric acid + magnesium 4) Hydrogen peroxide with catalyst Popular. - ppt download

balance the equation Sodium bicarbonate + Sulphuric acid = Sodium sulphate + Water + Carbon dioxide. - Brainly.in
Sodium carbonate reacts with dil. H2SO4 to give the respective salt, water and carbon dioxide. - Sarthaks eConnect | Largest Online Education Community
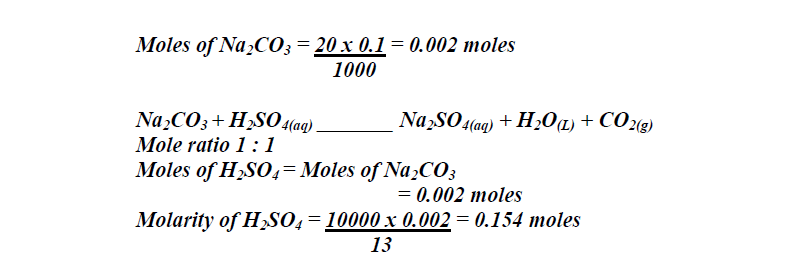
In a reaction 20cm<sup>3</sup> of 0.1 M Sodium Carbonate completely reacted with 13cm<sup>3</sup> of dilute sulphuric acid. Find the molarity of the sulphuric acid used.

Describe the following chemical reaction giving their balance equation Sodium carbonate is treated with dilute sulphuric acid - Science - Carbon An Important Element - 14423557 | Meritnation.com