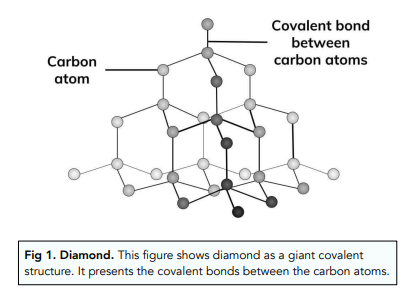
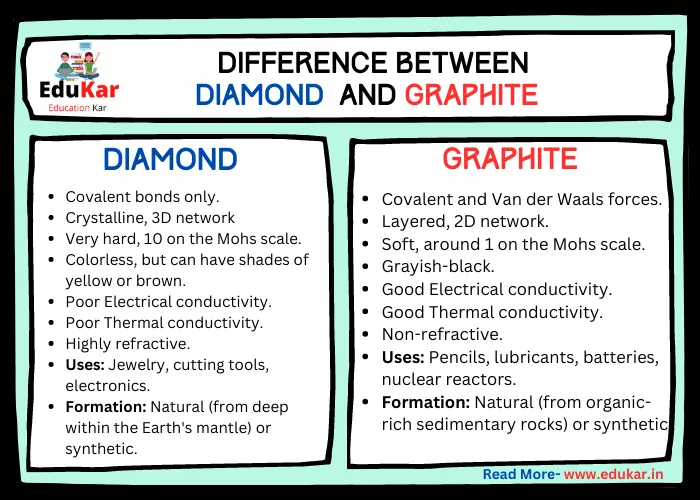
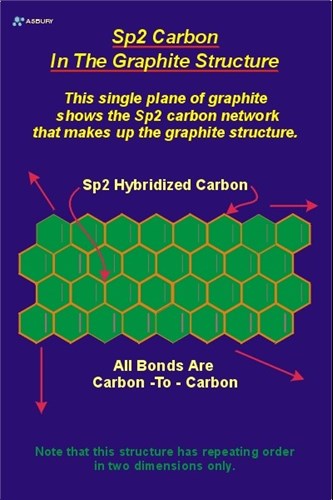
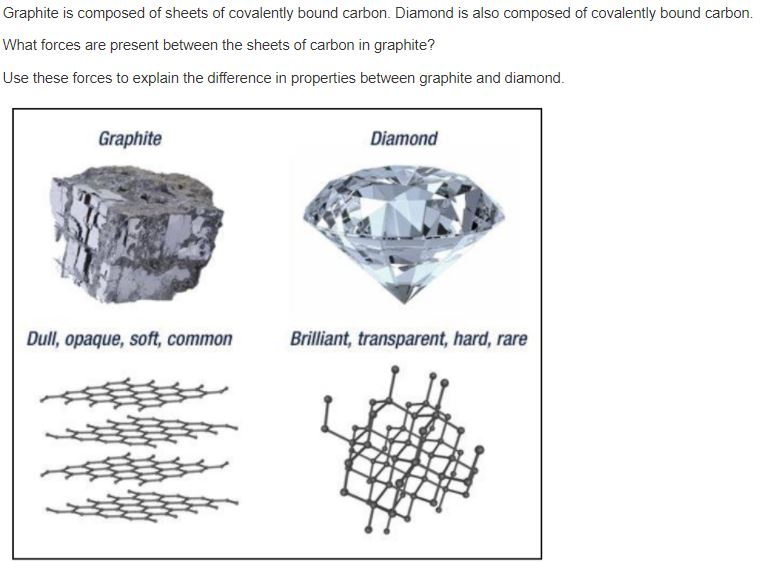
Explain the difference between diamond and graphite that what makes the graphite opaque to light while diamond is transparent to - Science - Carbon and its Compounds - 10691577 | Meritnation.com

The critical role of carbon in marrying silicon and graphite anodes for high‐energy lithium‐ion batteries - Wu - 2019 - Carbon Energy - Wiley Online Library











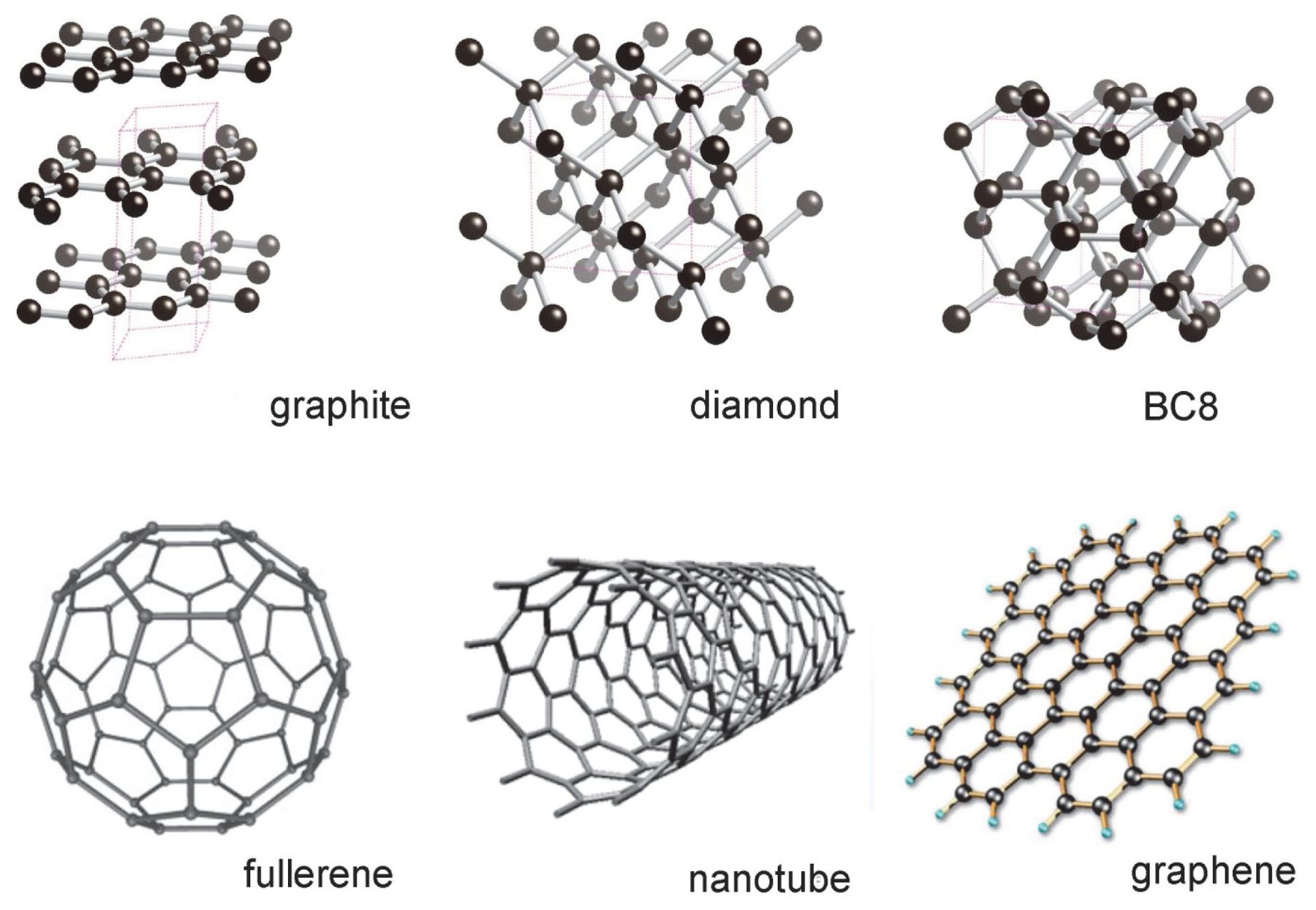





.jpg)