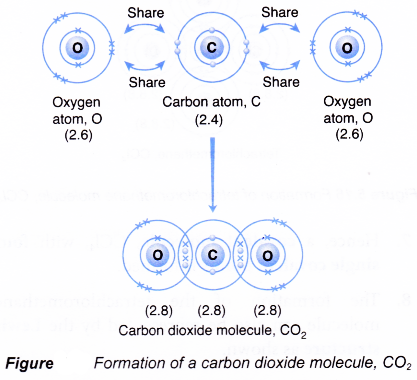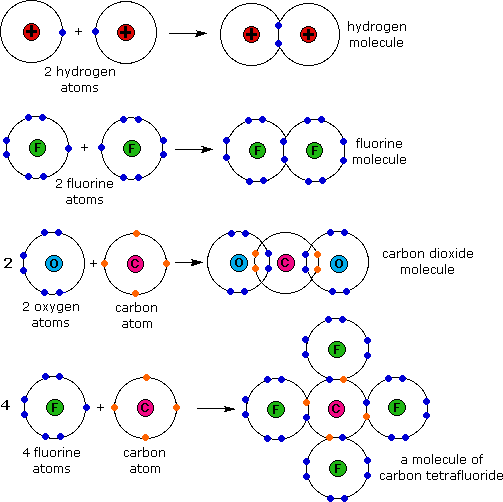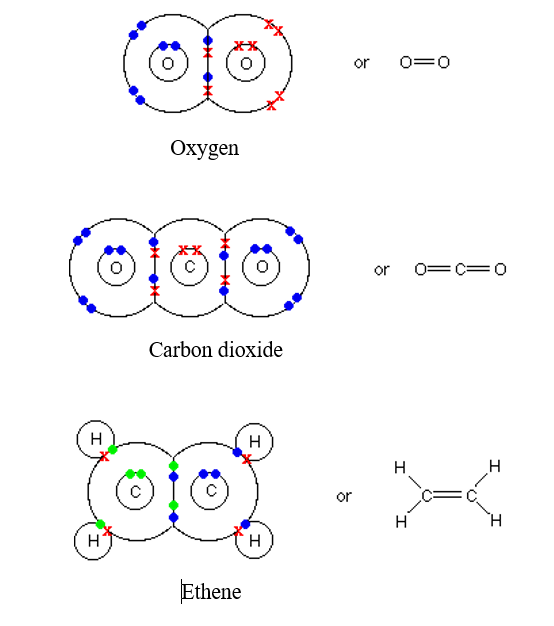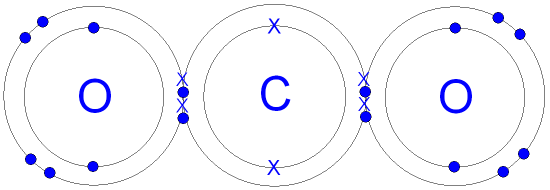
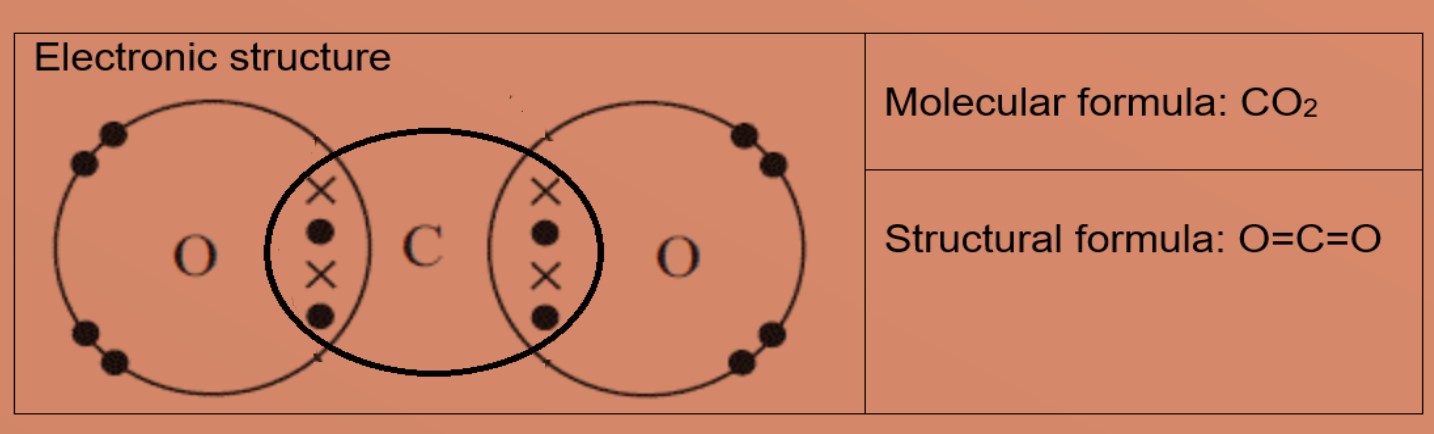
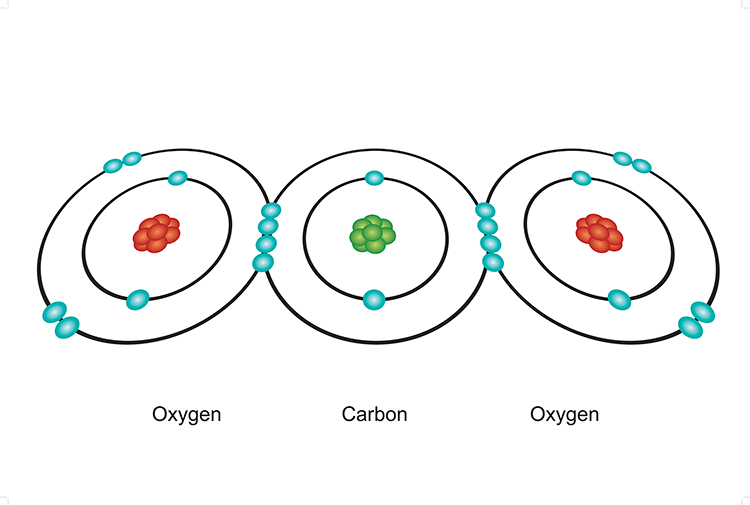
GCSE CHEMISTRY - Covalent Bonding in a Carbon Dioxide Molecule - What is the Structure of a Carbon Dioxide Molecule? - GCSE SCIENCE.
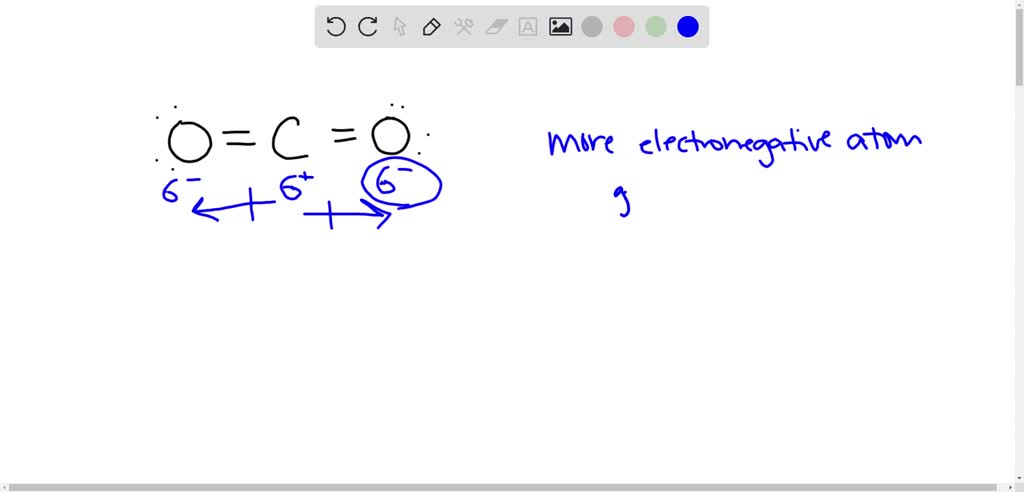
SOLVED: The bonds between carbon and oxygen in carbon dioxide are polar covalent bonds. In these bonds, the oxygen atom is assigned a δ − sign because. the carbon atom is larger

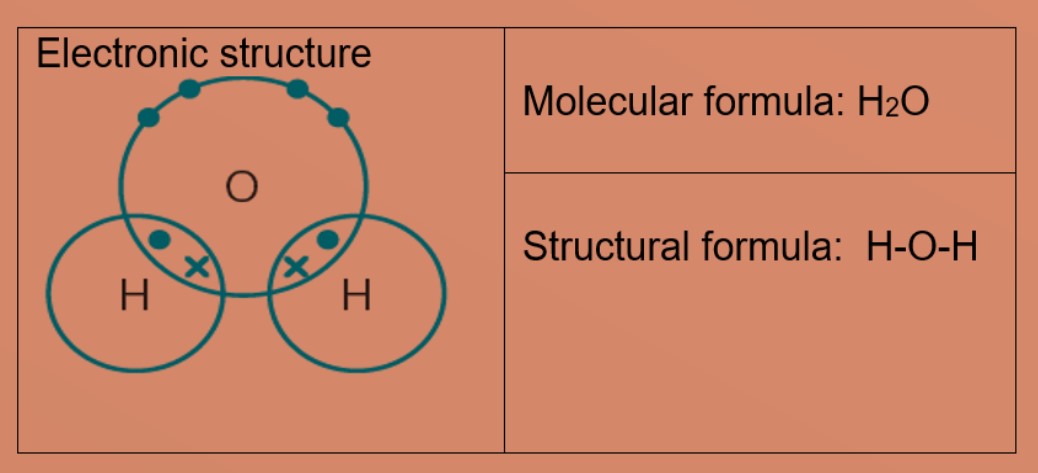
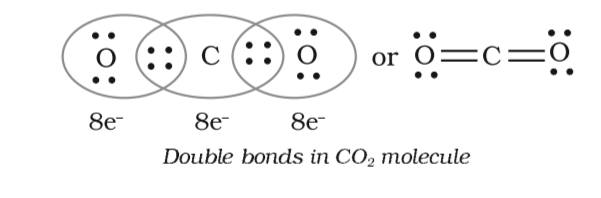
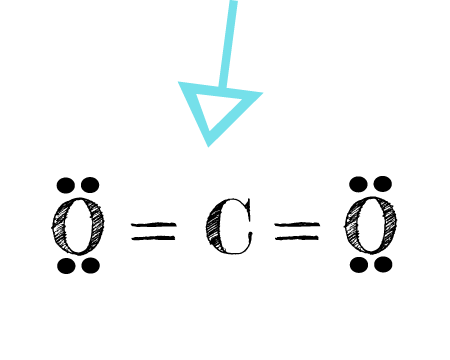
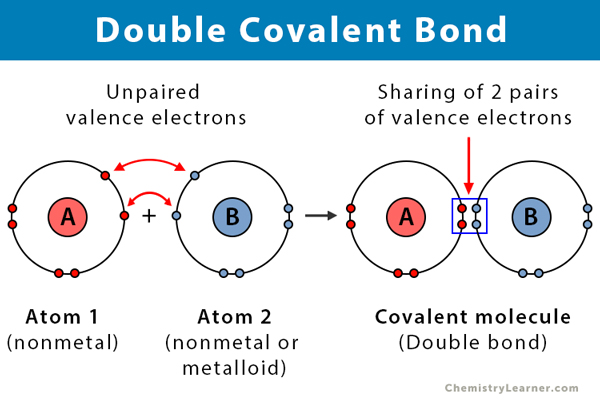
Draw the electron dot structures of the following compounds and state the type of bonding in each case: i CO2 ii MgO iii H2O iv HCI v MgCl2







:max_bytes(150000):strip_icc()/nitrogen-molecule-122373927-57d6b2463df78c58336b37f5.jpg)