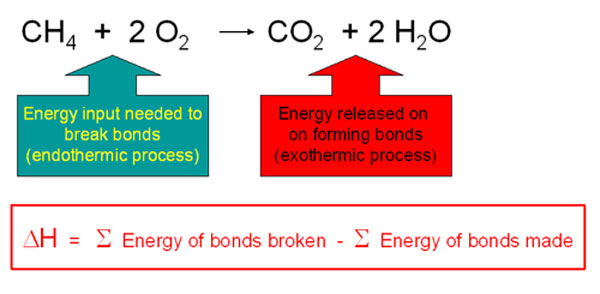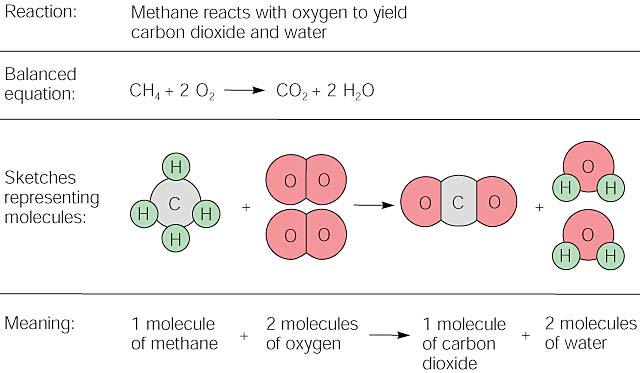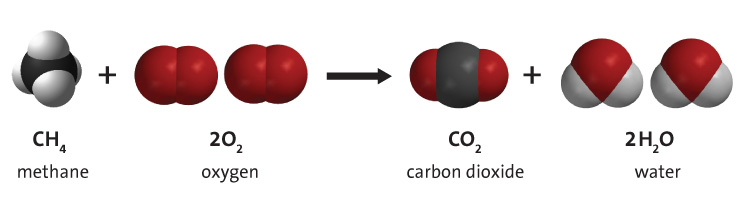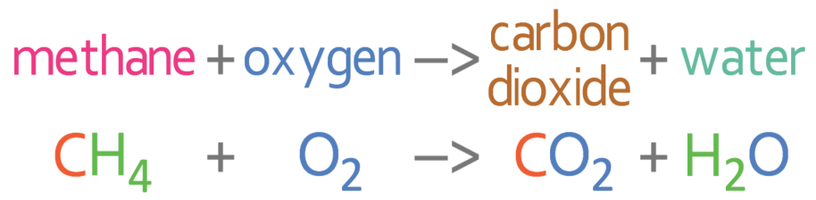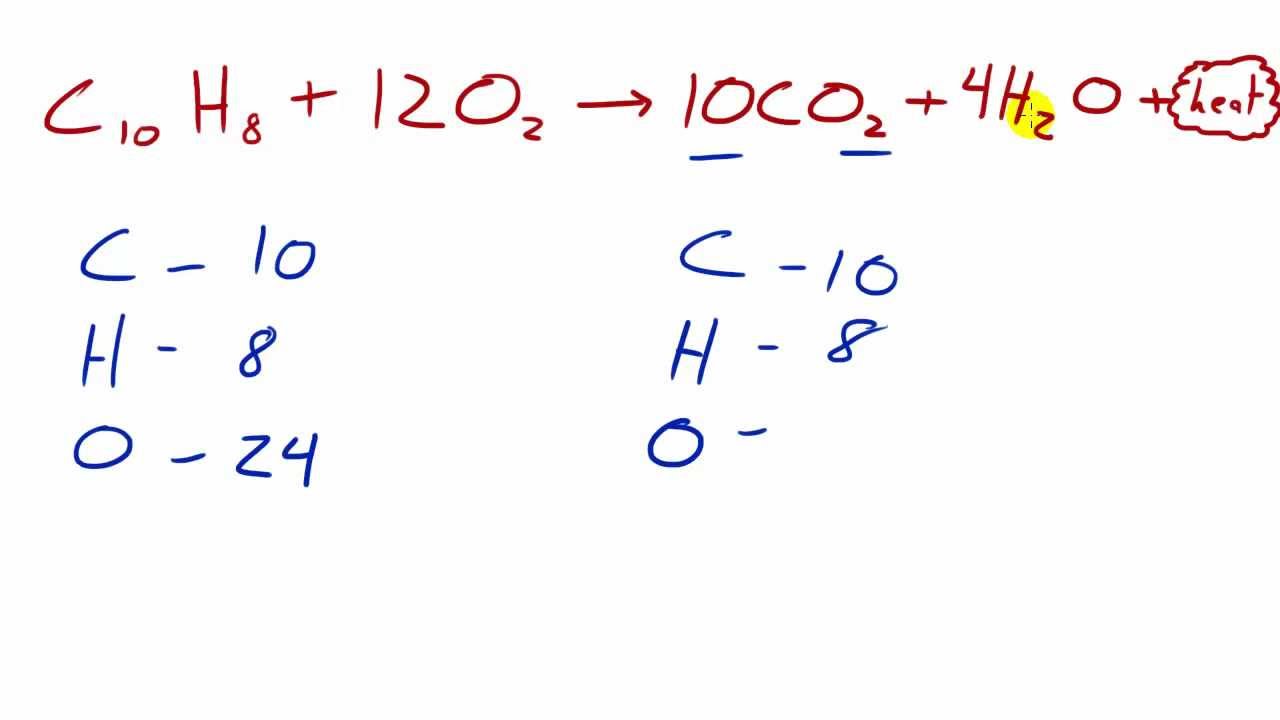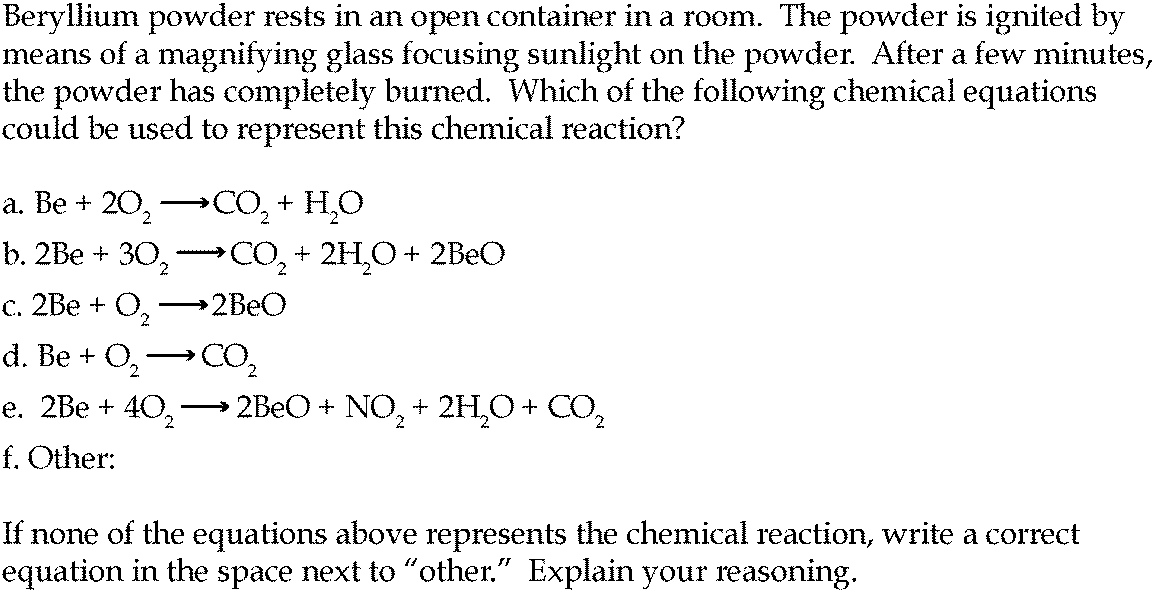
Combustion always produces carbon dioxide and water”: a discussion of university chemistry students' use of rules in place of principles - Chemistry Education Research and Practice (RSC Publishing) DOI:10.1039/C4RP00089G
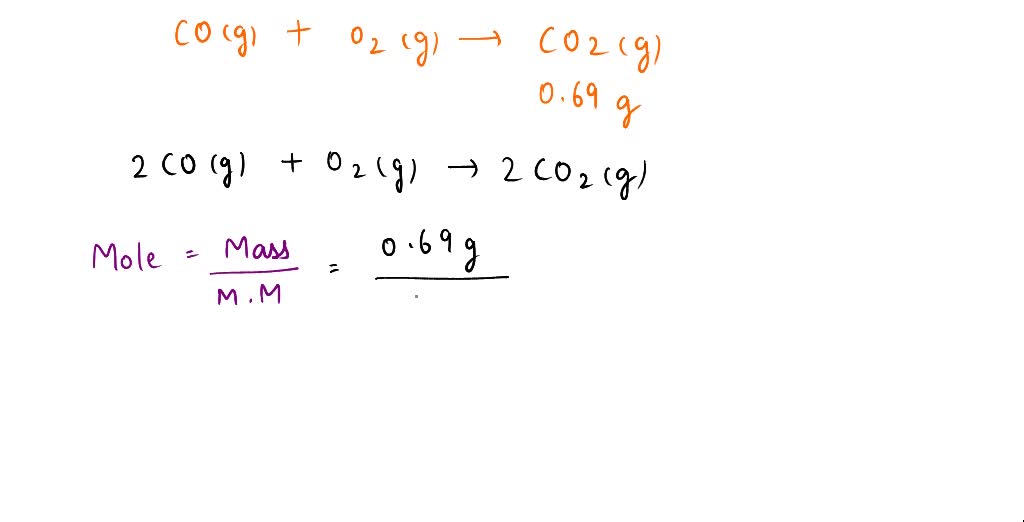
SOLVED: In the combustion of carbon monoxide, what mass of CO is required to produce 0.69 grams of carbon dioxide? The unbalanced equation is shown below: CO (g) + O2 (g) —–> CO2 (g)

Write the balanced chemical equation for combustion of carbon dioxide - Science - Chemical Reactions and Equations - 12555075 | Meritnation.com