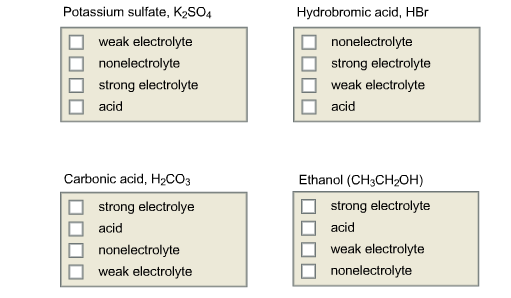
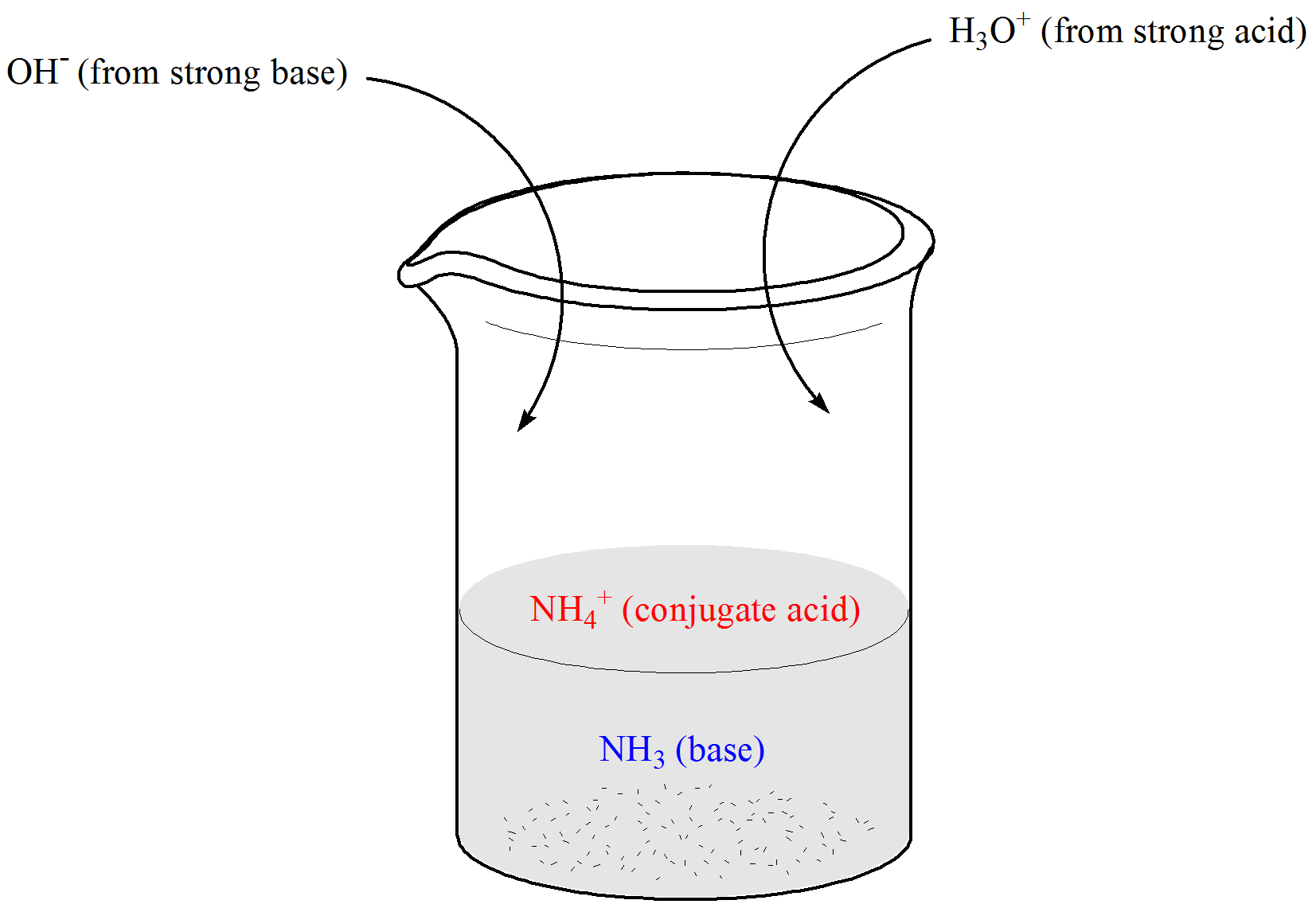
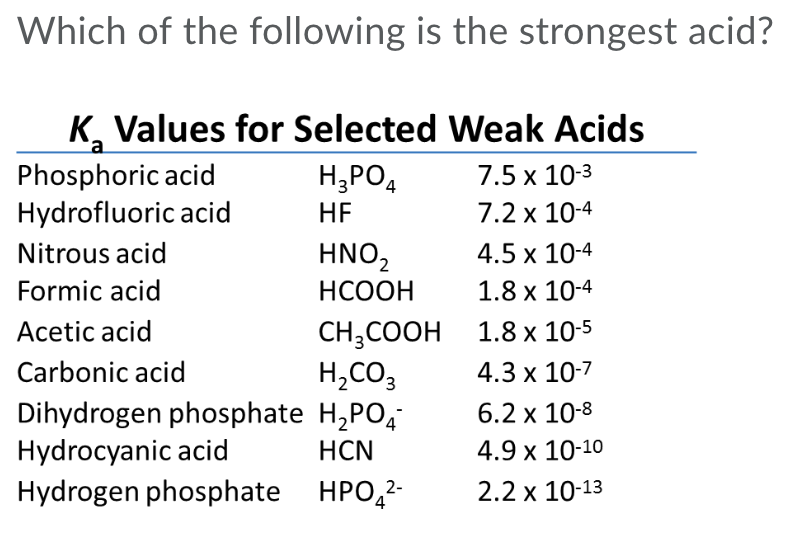
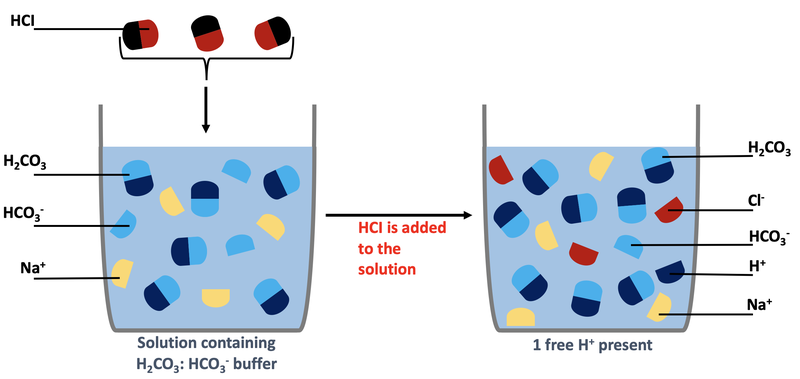
Why is carbonic acid a weak acid even though it gets completely dissociated into H+ and CO3- ions? - Quora

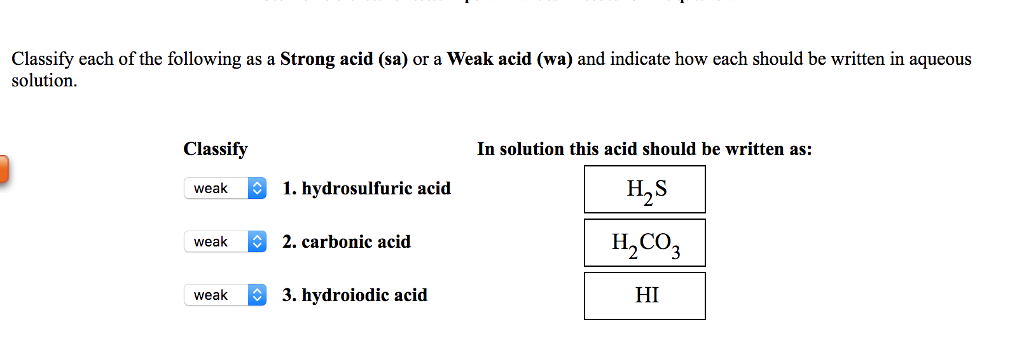
SOLVED: Classify each of the following aS a Strong acid (sa) O a Weak acid (wa) and indicate how each should be written in aqueous solution: Classify In solution this acid should




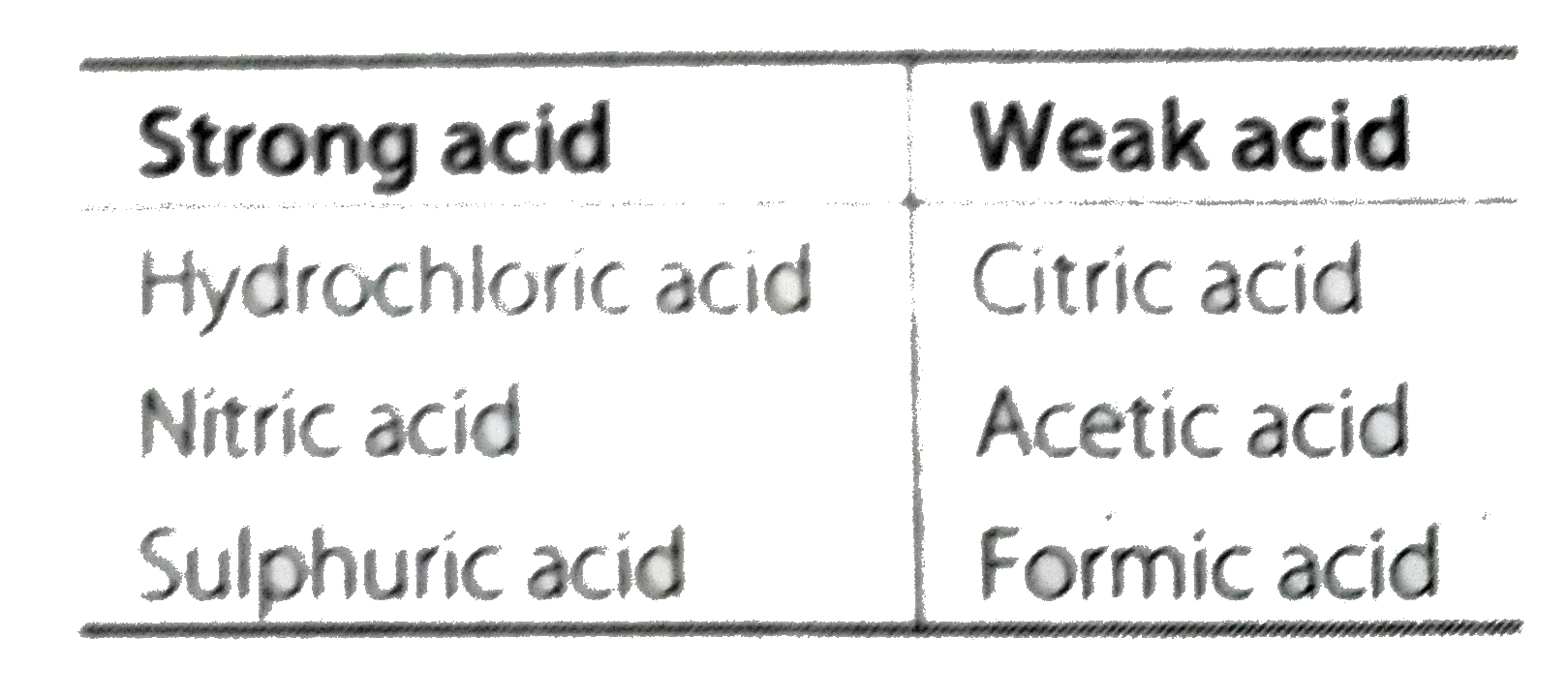
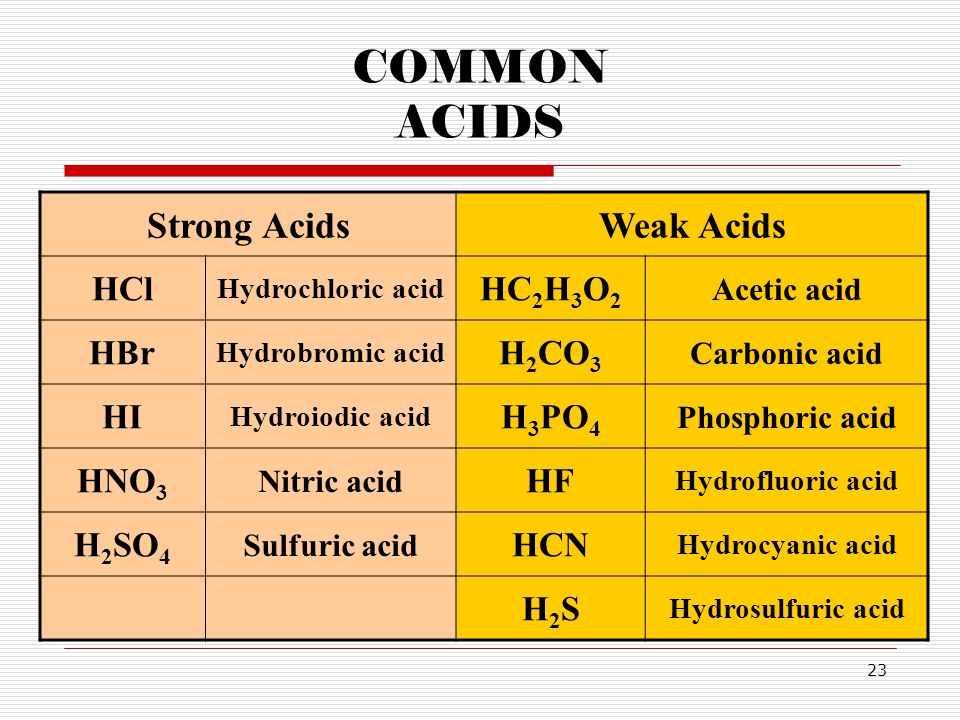

:max_bytes(150000):strip_icc()/list-of-strong-and-weak-acids-603642-v2copy2-5b47abd0c9e77c001a395e55.png)