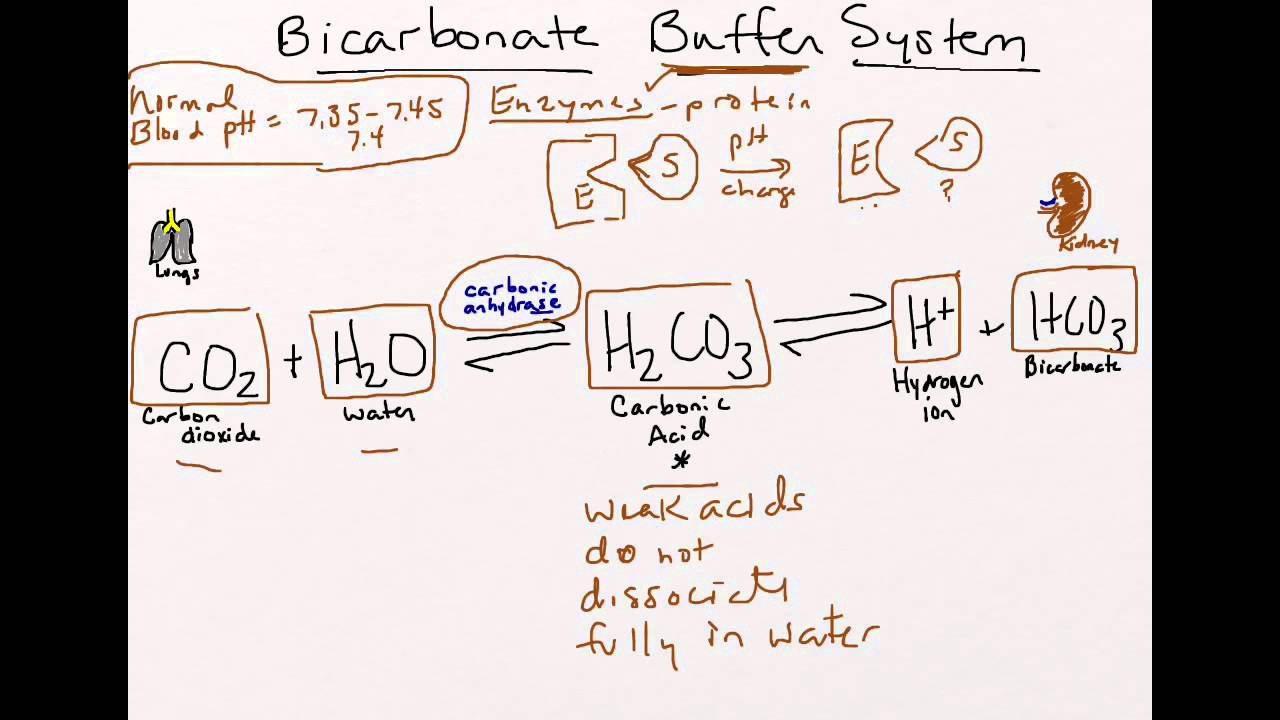
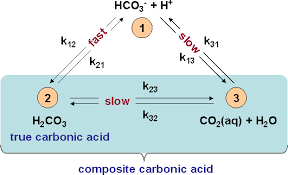
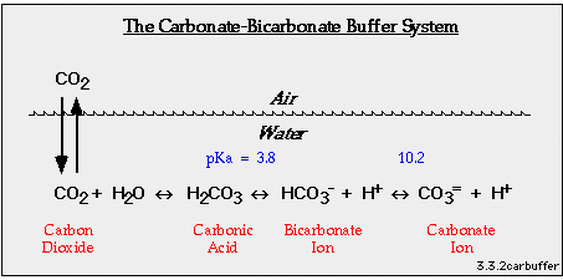
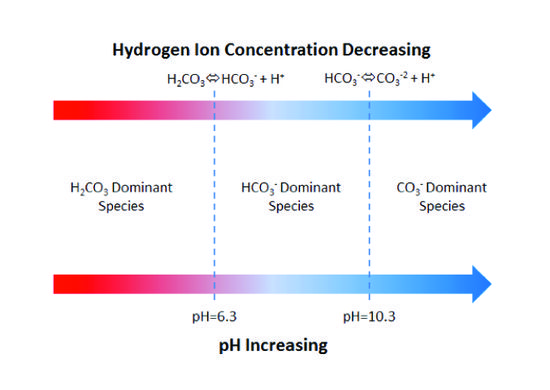
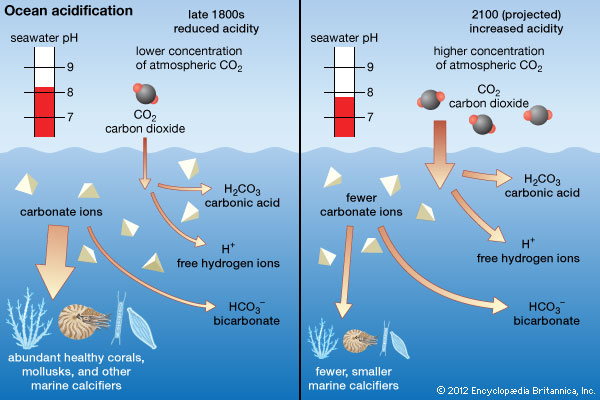
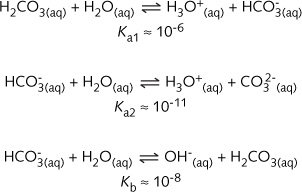Climate Change Education Across the Curricula, Across the GlobeVideo Micro-lecture: Ocean Buffer Chemistry

SOLVED: Application 2 The human body utilises a carbonate/hydrogen carbonate buffer system to help regulate the pH of blood and transport dissolved carbon dioxide, as shown by the following equation: HzOu) COzlaq)