Nitrogen-Doped Carbon Nanodots-Ionogels: Preparation, Characterization, and Radical Scavenging Activity | ACS Nano
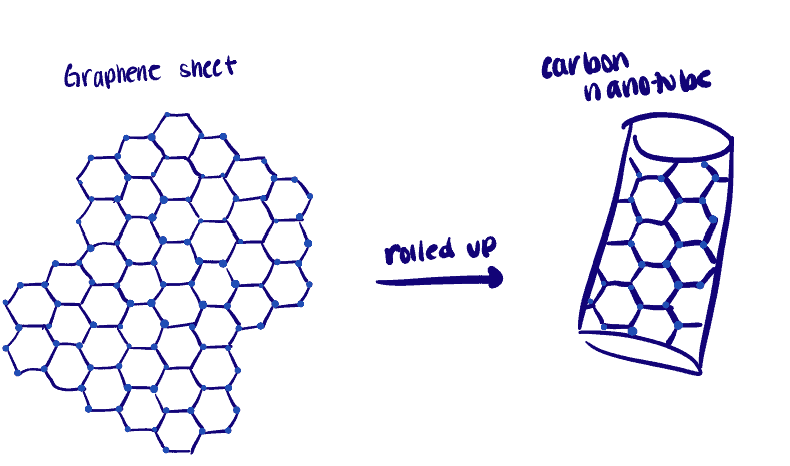
Synthesis and Applications of Nanocarbons: Properties of Carbon Bulk Materials: Graphite and Diamond

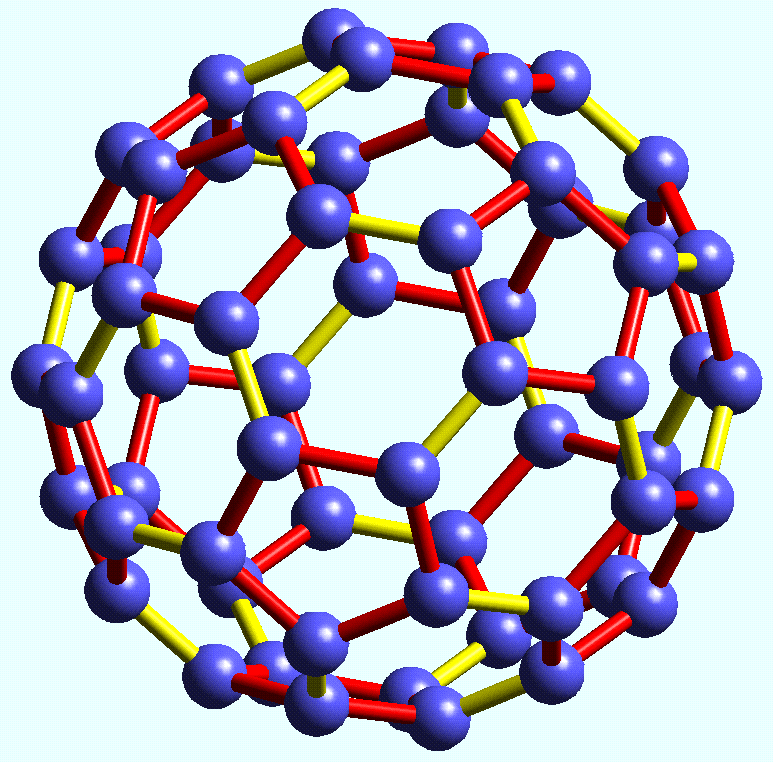
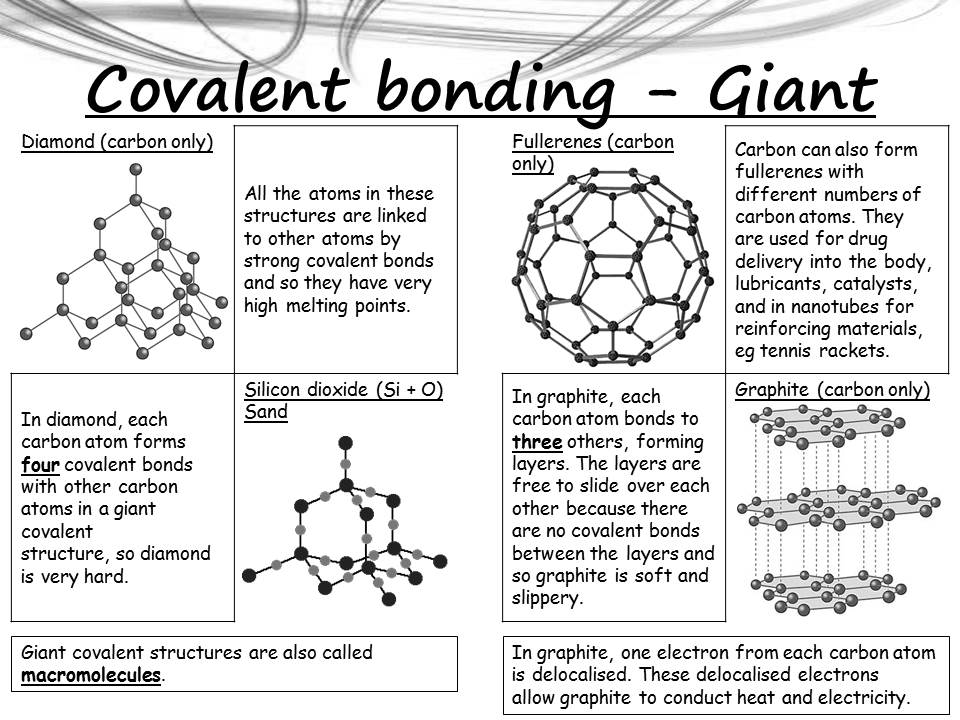
GCSE EDUQAS Chemistry Bonding , structure and properties Complete Revision Summary - Expert Guidance By Mahima Laroyia











.jpg)