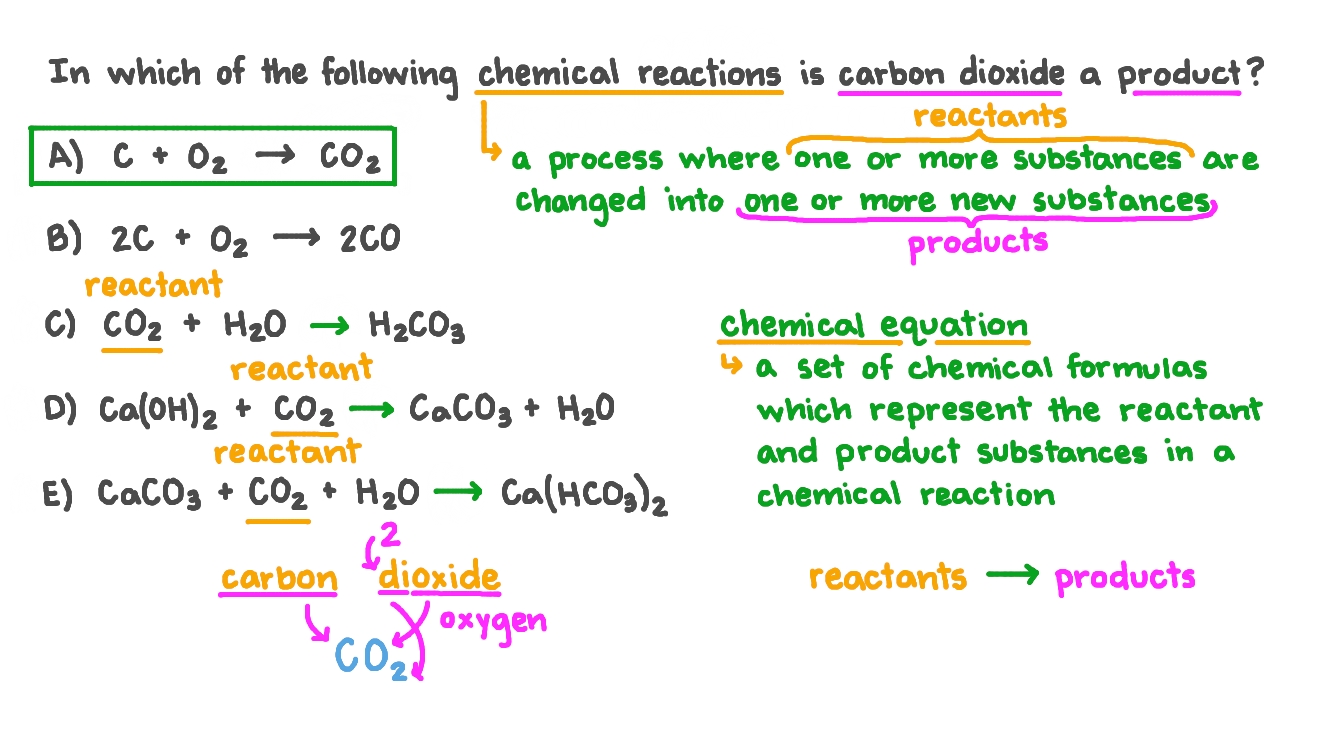
.png)
how will carbon react with oxygen what happens when the product is dissolved in water show along with - Science - Materials Metals and Non-Metals - 1217495 | Meritnation.com

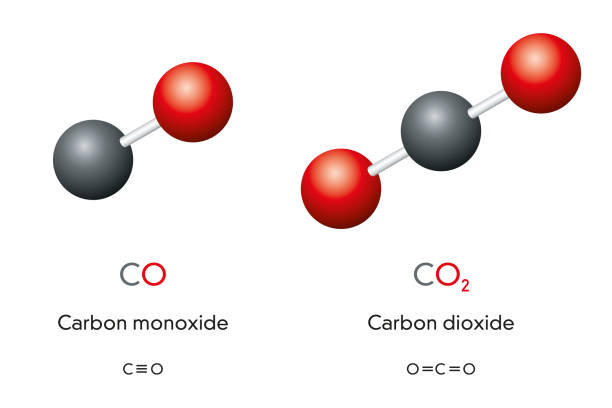
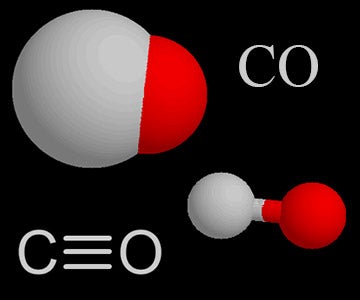
The heat of the combustion of graphite and carbon monoxide respectively are 393.5 kJ mol1 and 283 kJ mol ^-1 . Thus heat of formation of carbon monoxide in kJ mol ^-1 is:
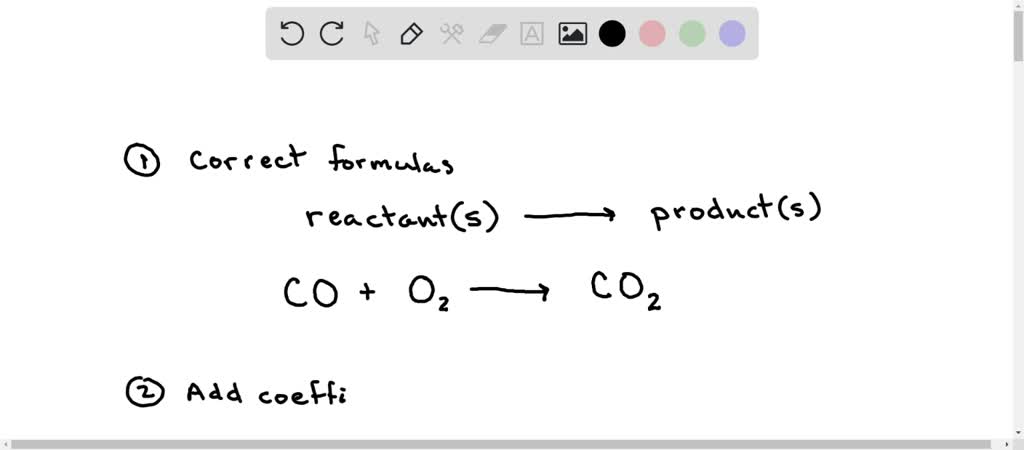
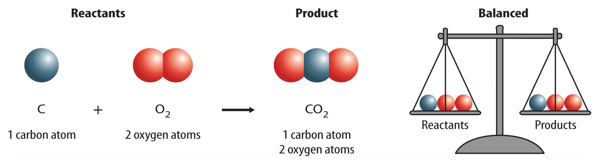
SOLVED: Write a balanced equation for the following reaction: When carbon monoxide combines with oxygen, carbon dioxide is formed. (Use the lowest possible coefficients. Omit states of matter.) Reactants Products + Step

Hydrogen, oxygen and carbon dioxide are taken in containers of 2 l volume each. Compare the number of molecules of the three gases under same conditions of temperature and pressure.
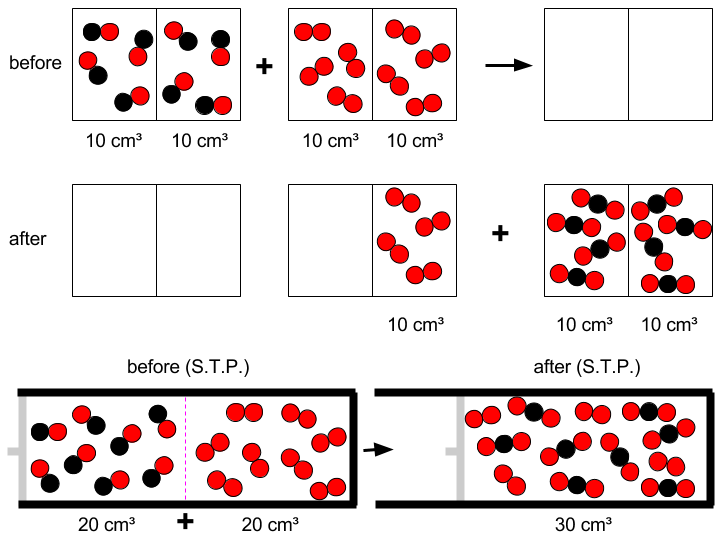
20*cm^3 volumes EACH of carbon monoxide and oxygen gas are combined and combusted. What volume of gas remains AFTER the reaction? | Socratic