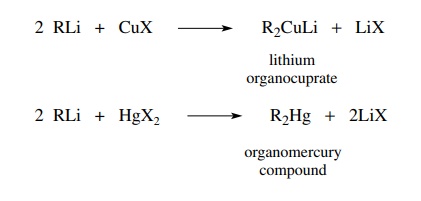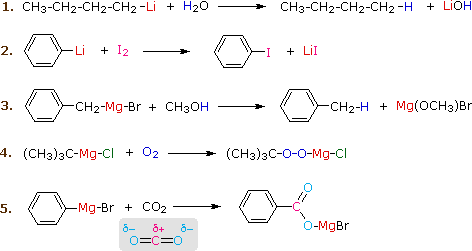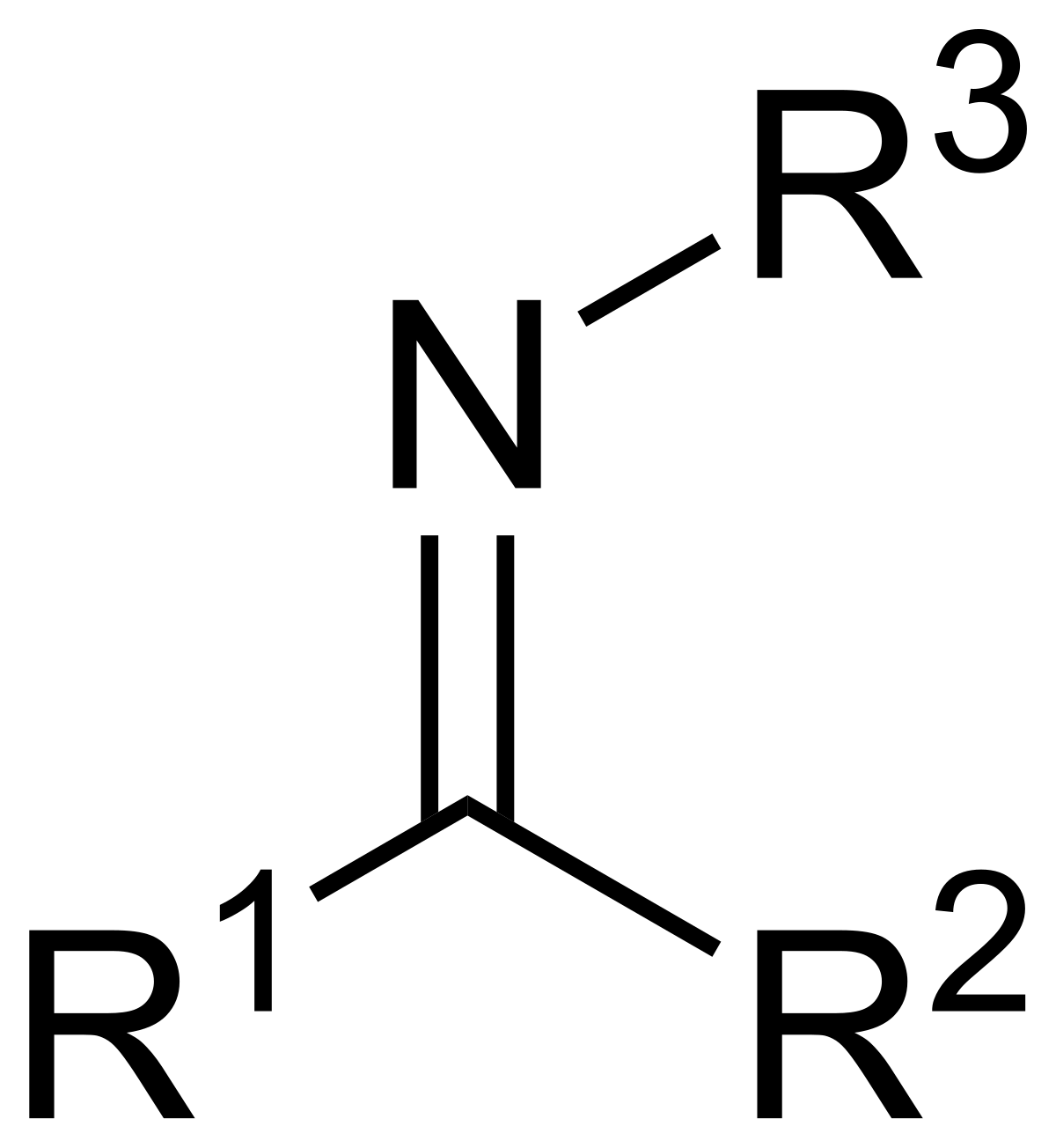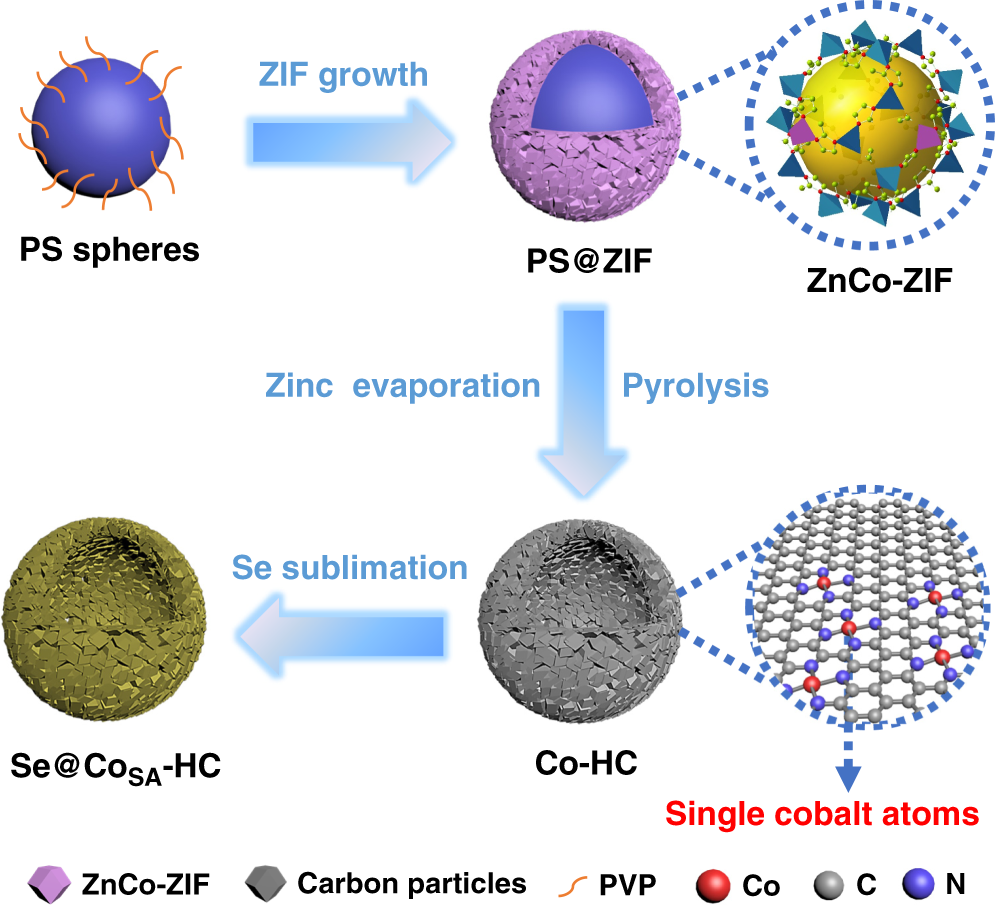
High-power lithium–selenium batteries enabled by atomic cobalt electrocatalyst in hollow carbon cathode | Nature Communications

High sulfur-containing carbon polysulfide polymer as a novel cathode material for lithium-sulfur battery | Scientific Reports

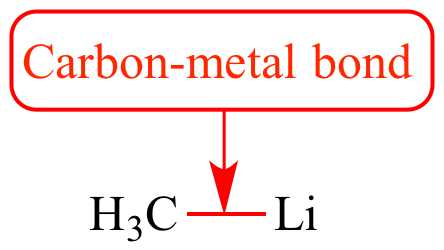
PDF) The Carbon-Lithium Electron Pair Bond in (CH3Li) n (n= 1, 2, 4) | Célia Fonseca Guerra - Academia.edu
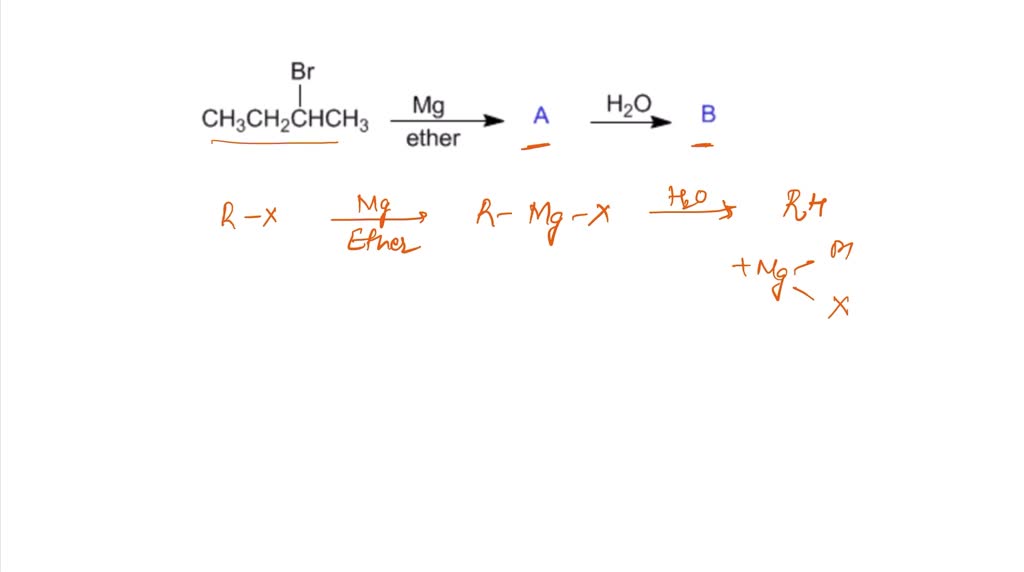
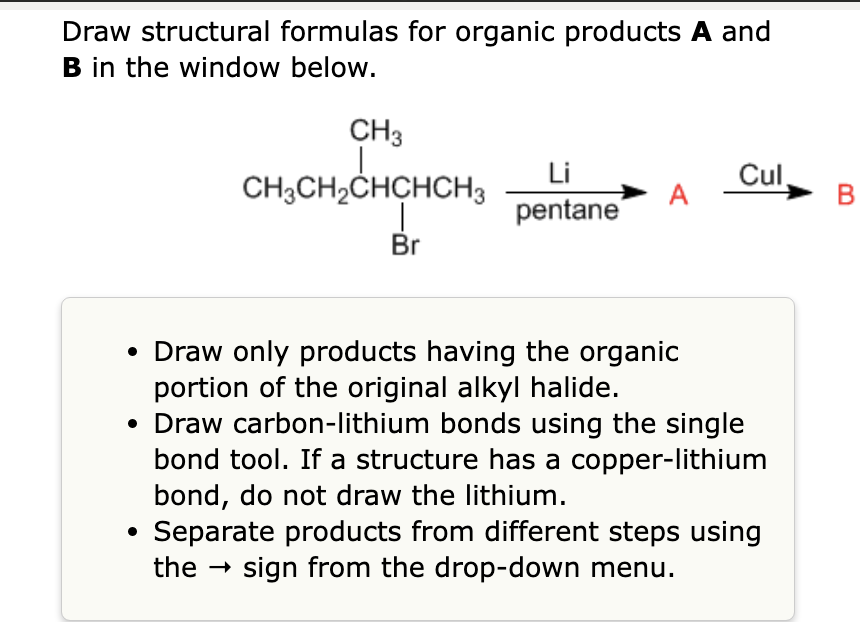
SOLVED: Draw the major organic product of the following reaction Mg ether Ho A? B? Draw carbon-lithium or carbon-copper bonds using the single bond tool If a structure has copper-lithium bond draw
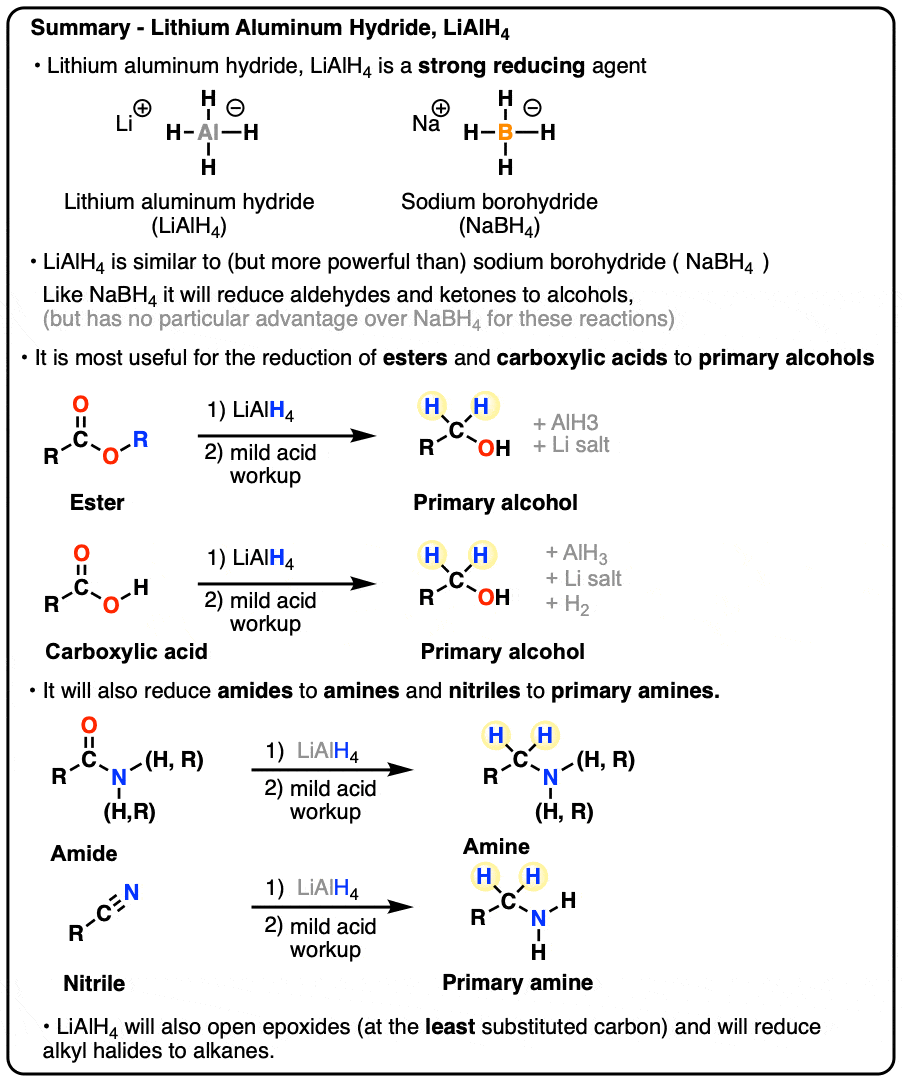
Lithium Aluminum Hydride (LiAlH4) For Reduction of Carboxylic Acid Derivatives – Master Organic Chemistry
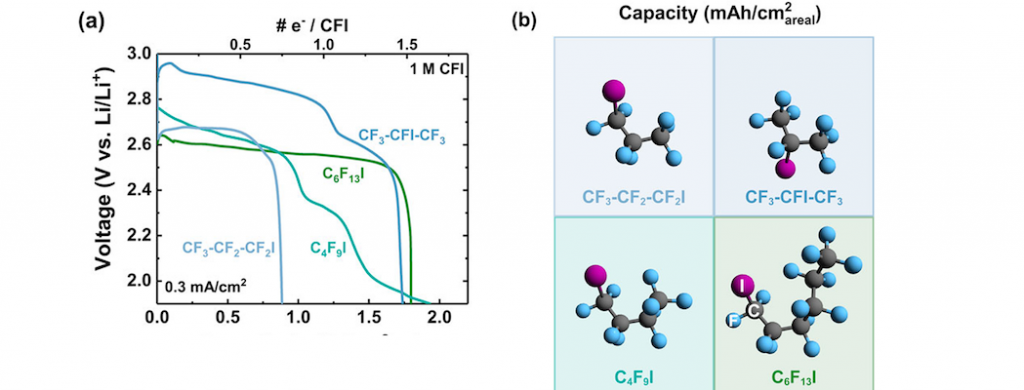
Lithium primary batteries employing multi-electron carbon-fluorine bond cleavage in perfluoroalkylated reactants | Gallant Energy and Carbon Conversion Lab

Lithium Enolates in the Enantioselective Construction of Tetrasubstituted Carbon Centers with Chiral Lithium Amides as Noncovalent Stereodirecting Auxiliaries | Journal of the American Chemical Society