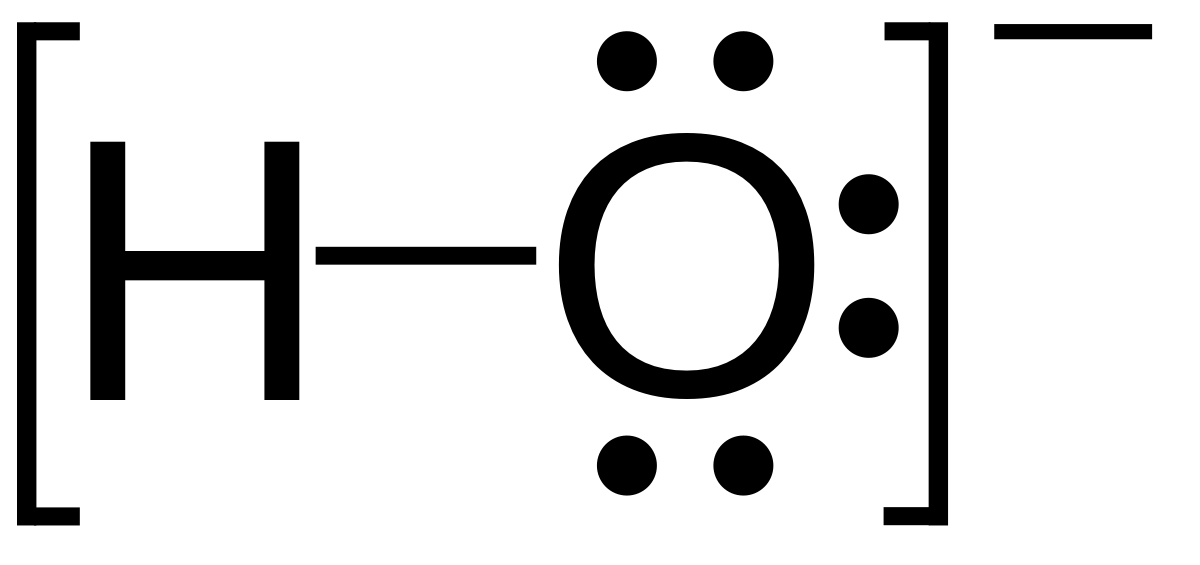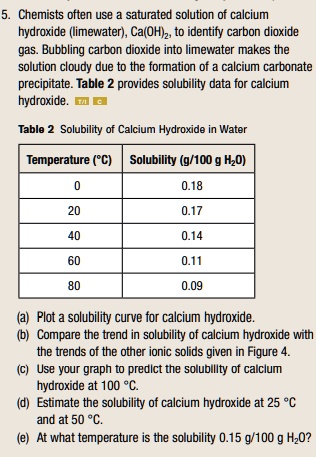
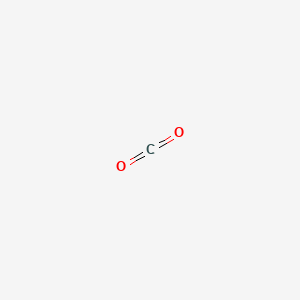
SOLVED: Chemists often use saturated solution of calcium hydroxide (limewater) , Ca(OHIz; to identify carbon dioxide gas. Bubbling carbon dioxide into limewater makes the solution cloudy due to the formation of calcium
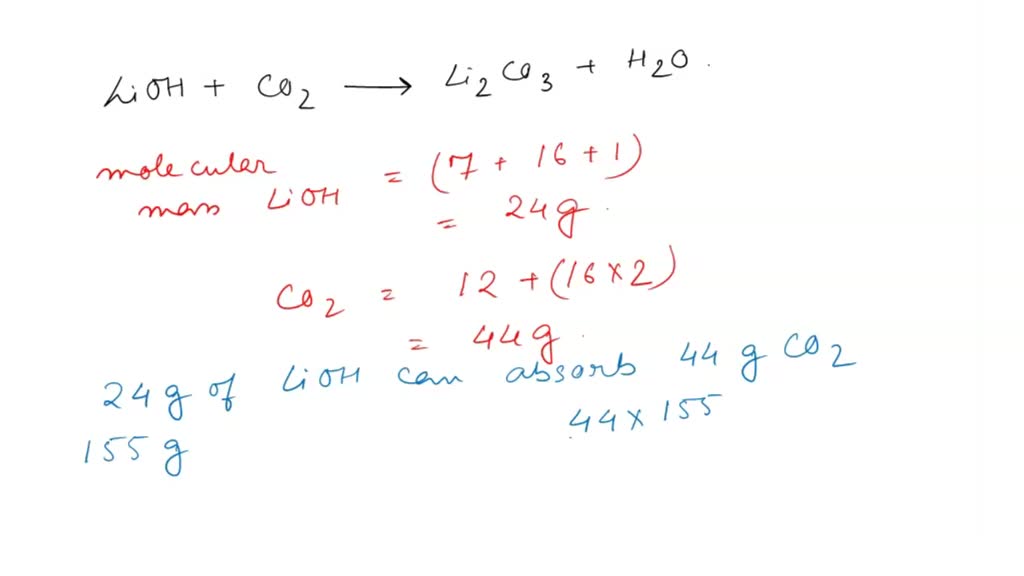
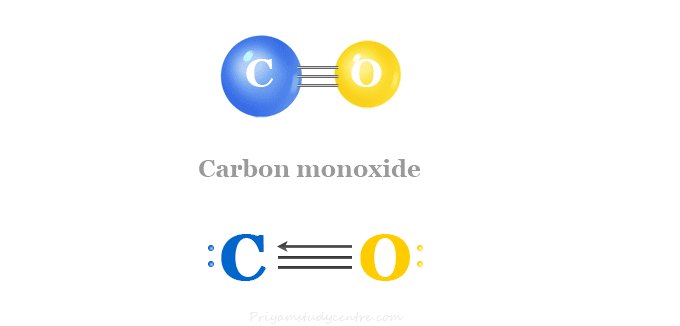
SOLVED: Alkali metal hydroxides are sometimes used to "scrub" excess carbon dioxide from the air in closed spaces (such as submarines and spacecraft). For example, lithium hydroxide reacts with carbon dioxide according
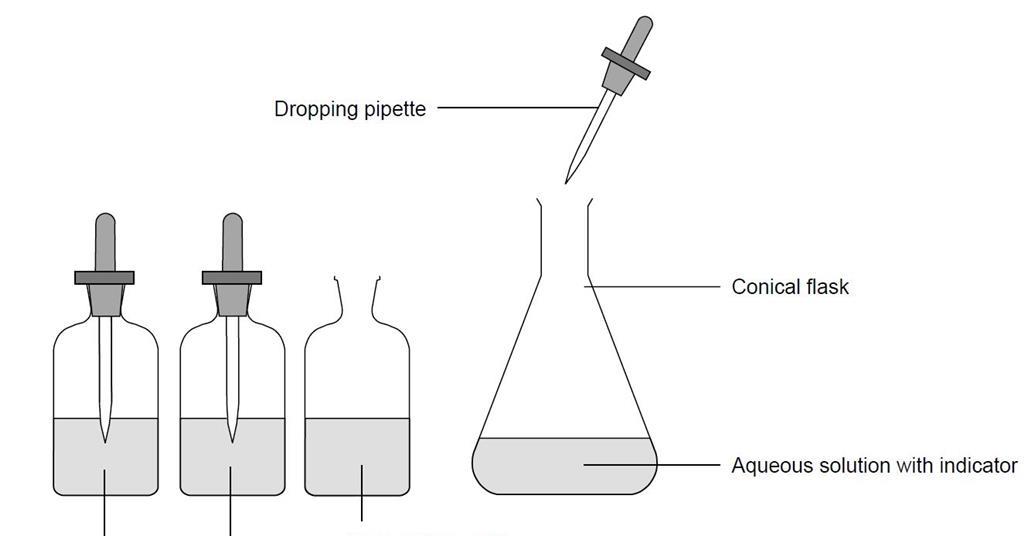
The diagram below represents part of a set-up used to prepare and collect gas T. (a) Name two - Tutorke

Uses Limestone cycle manufacture cement concrete glass clay ceramics thermal decomposition carbonates, nitrates, slaked lime calcium oxide hydroxide quarrying issues gcse igcse O level chemistry revision notes

Write the balanced chemical equations for the following reactions.A Calcium hydroxide + Carbon dioxide → Calcium carbonate + waterB Zinc + Silver nitrate → Zinc nitrate + SilverC Aluminium + copper chloride

SOLVED: Part The fermentation of sucrose by in an aqucous solution produces ethanol and carbon dioxide when anaerobic conditions are ysec; iccordiqg to the following unbalanced chemical equation: Zymascs C12H22011 Hz0 CH;CHzOH

Supported lithium hydroxide for carbon dioxide adsorption in water-saturated environments | Request PDF


.PNG)