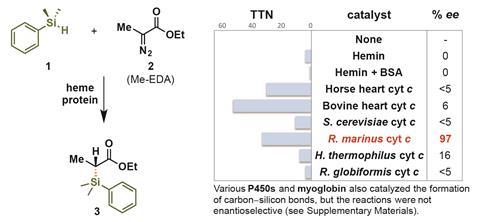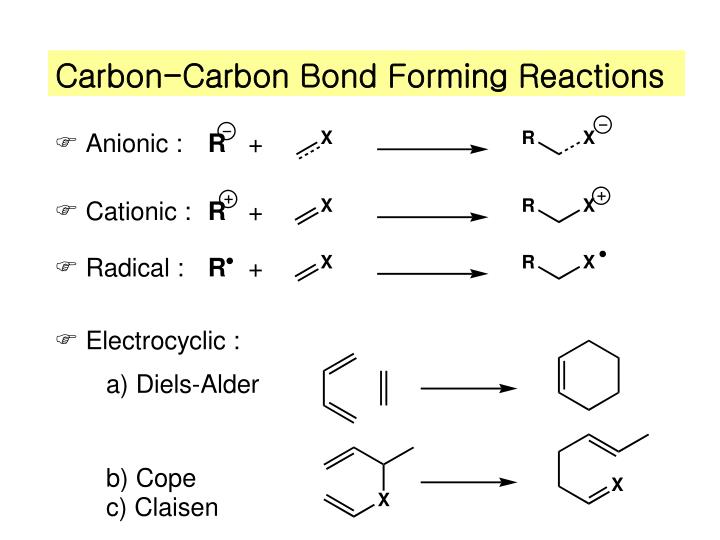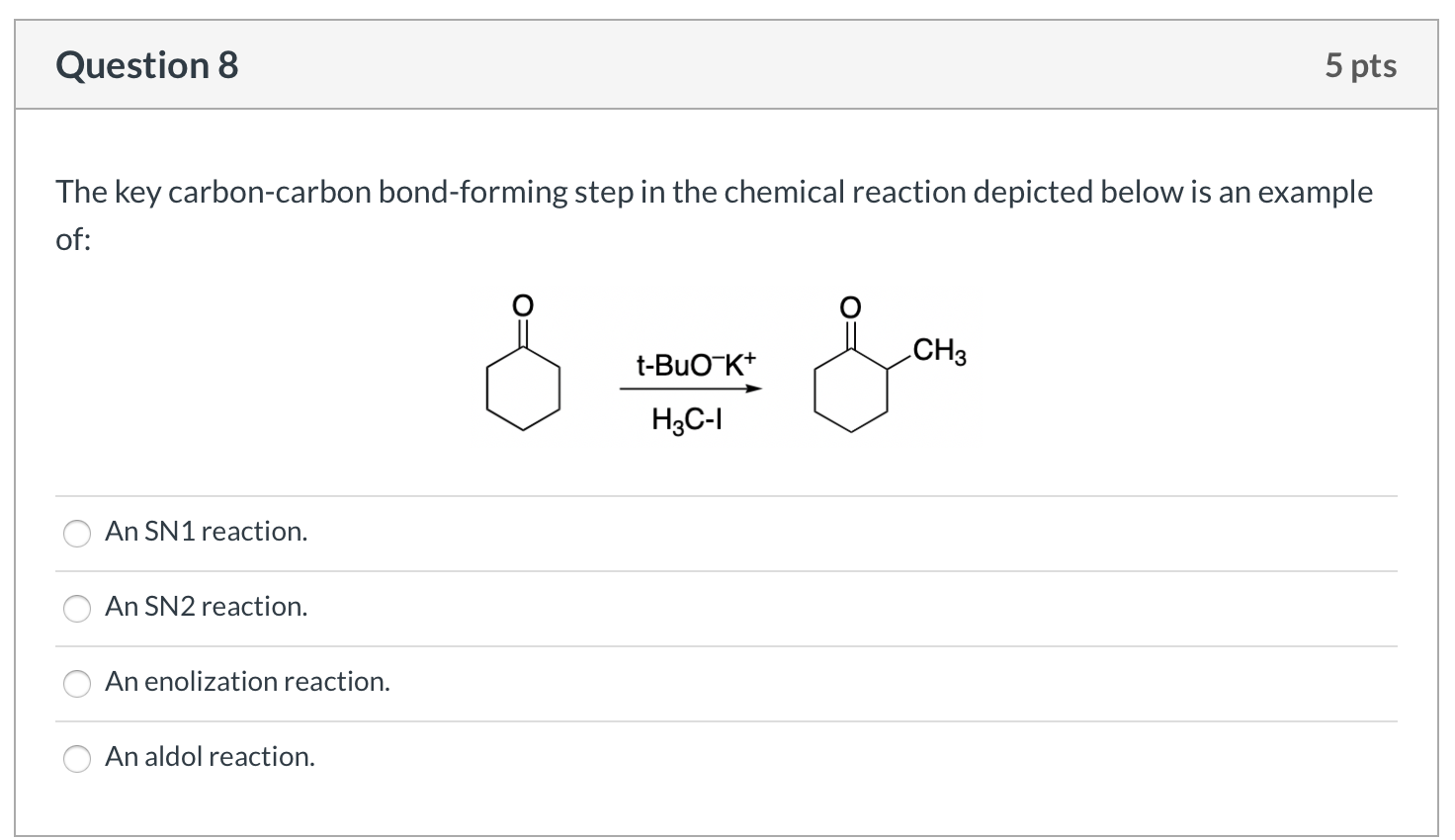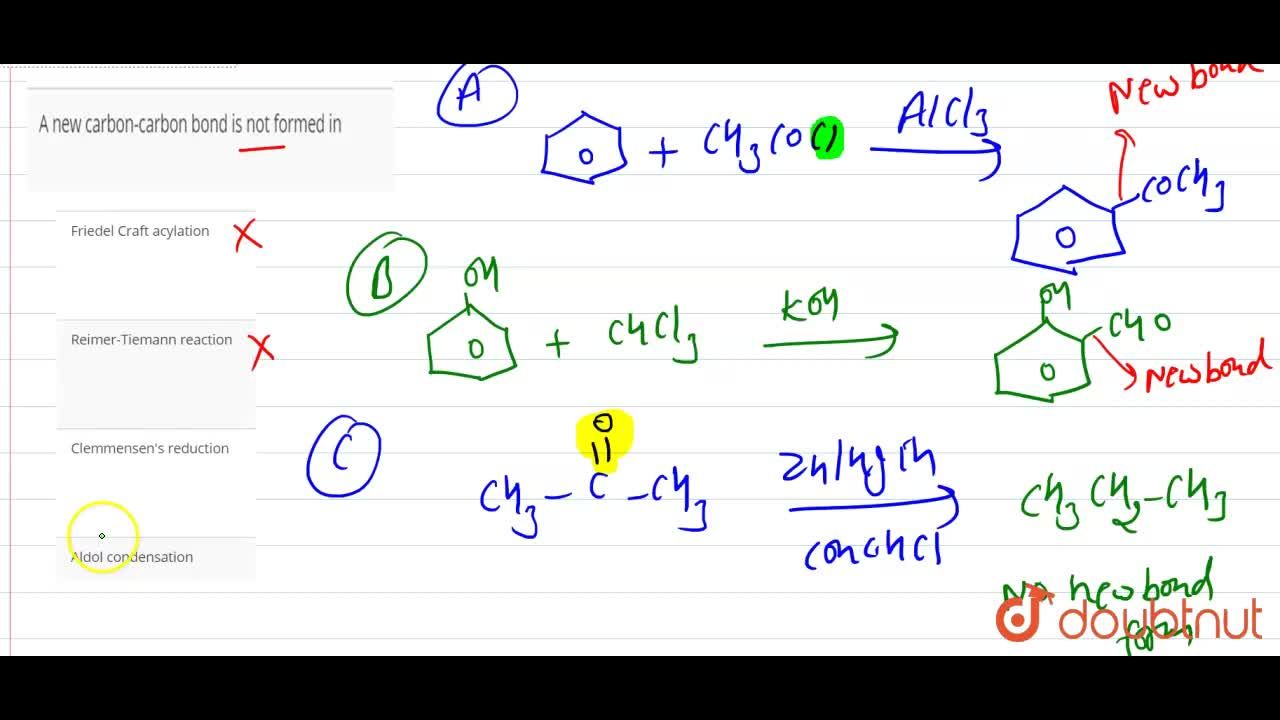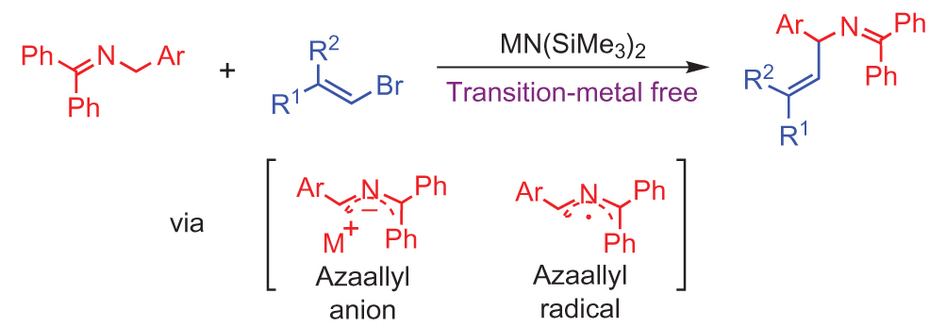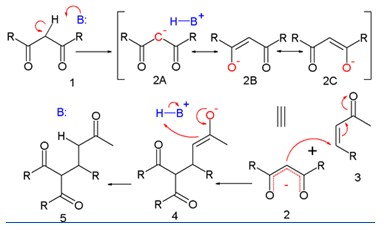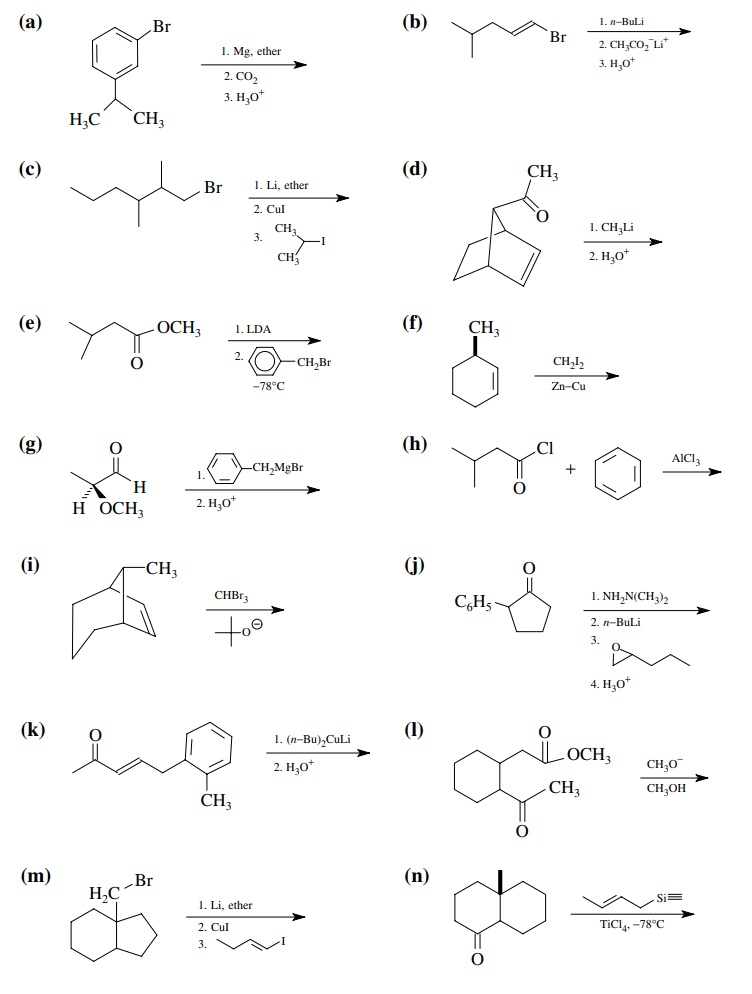
Solved Problems on Carbon-Carbon Bond Formation Between Carbon Nucleophiles and Carbon Electrophiles - Organic Chemistry
Design for carbon–carbon bond forming reactions under ambient conditions - RSC Advances (RSC Publishing) DOI:10.1039/C6RA14399G

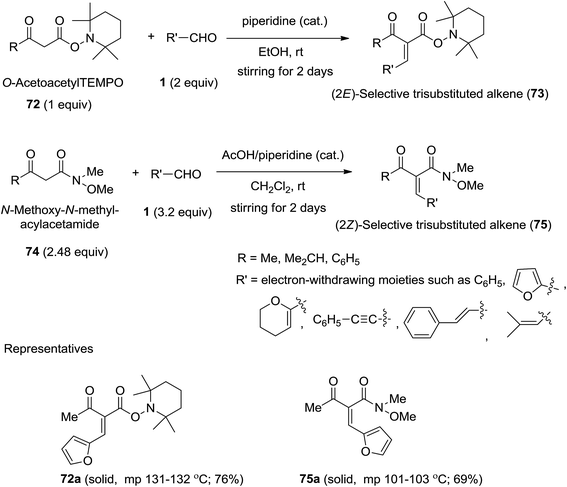
Stereodivergent Carbon–Carbon Bond Formation between Iminium and Enolate Intermediates by Synergistic Organocatalysis | Journal of the American Chemical Society

Practical carbon–carbon bond formation from olefins through nickel-catalyzed reductive olefin hydrocarbonation | Nature Communications
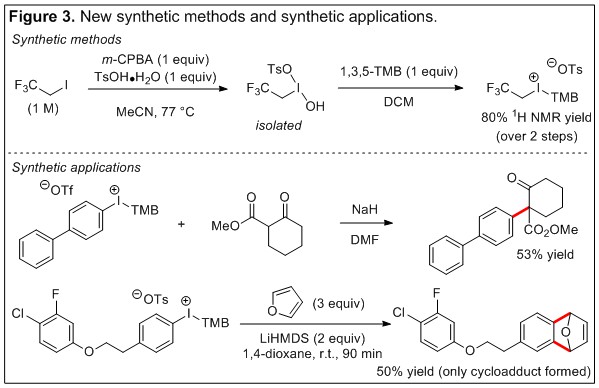
Report: Direct Carbon-Carbon Bond Forming Reactions with Hypervalent Iodine Reagents: Redefining Formal SNAr Reactivity with Carbon Nucleophiles (60th Annual Report on Research Under Sponsorship of The American Chemical Society Petroleum Research Fund)

Carbon–carbon bond activation of cyclobutenones enabled by the addition of chiral organocatalyst to ketone | Nature Communications
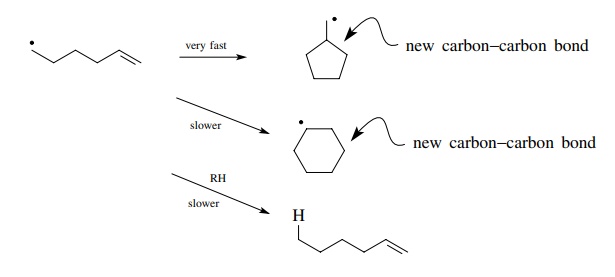
Nonpolymerization Reactions - Carbon-Carbon Bond Formation By Free-Radical Reactions | Organic Chemistry