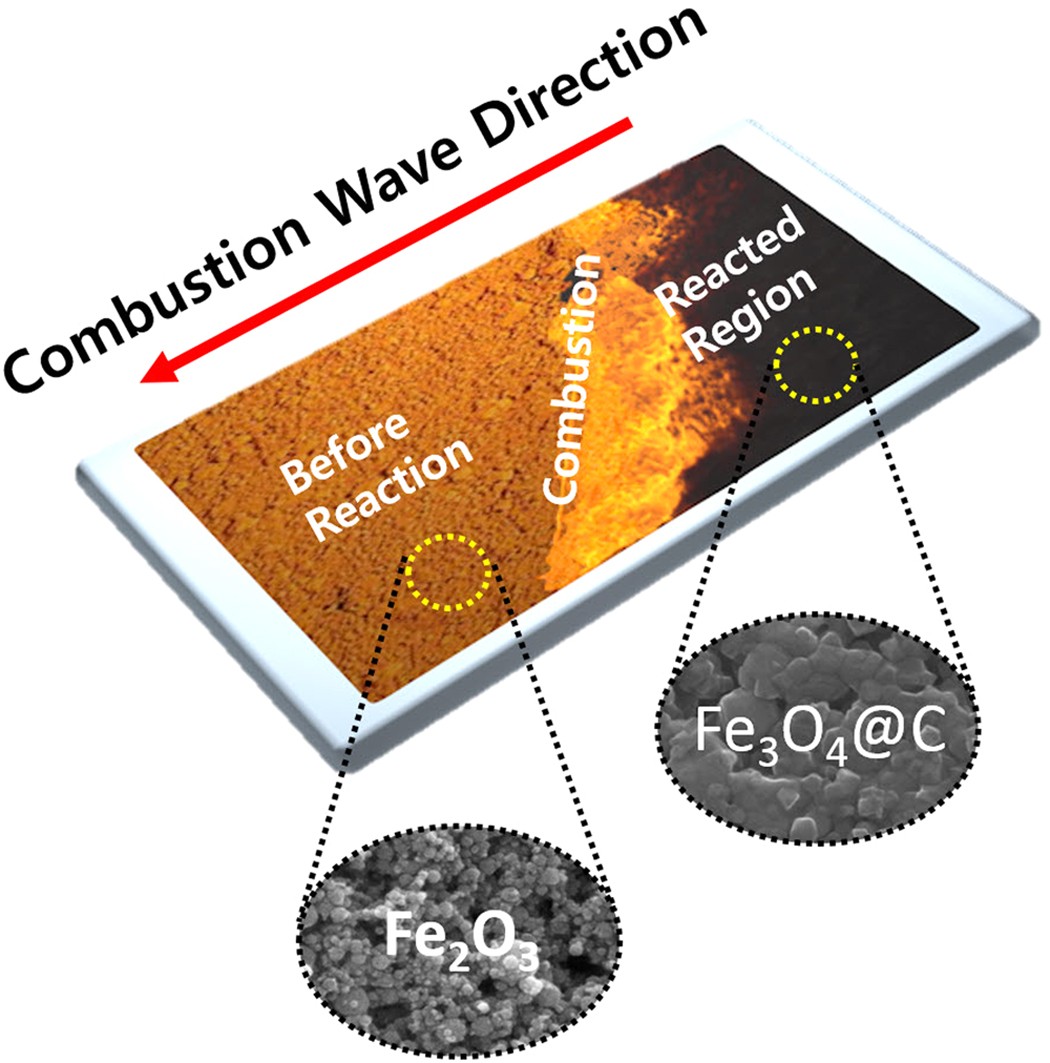
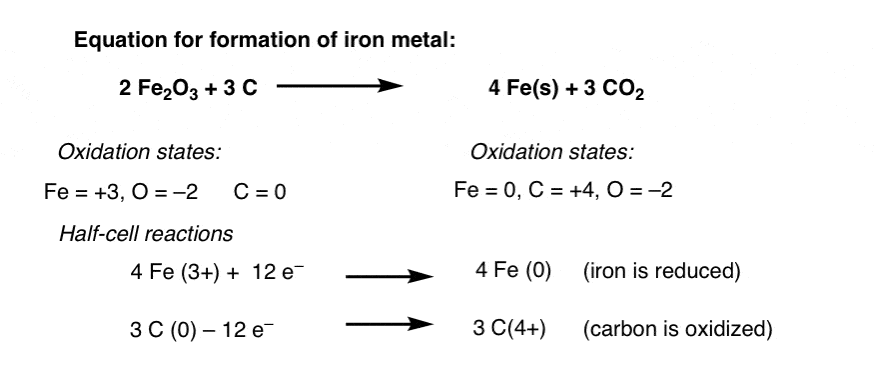
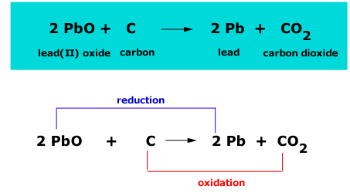
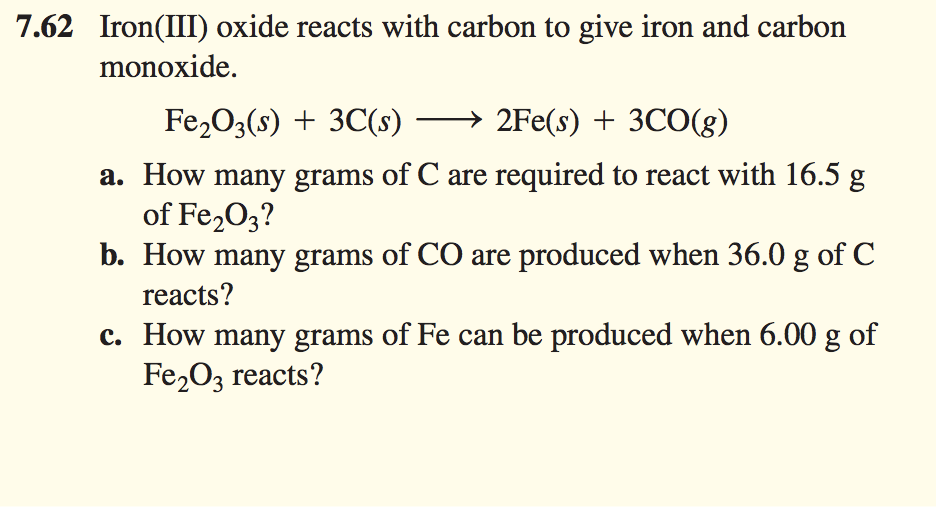a) Possible pathways to reduce iron (Fe) from iron oxide (Fe 2 O 3 or... | Download Scientific Diagram
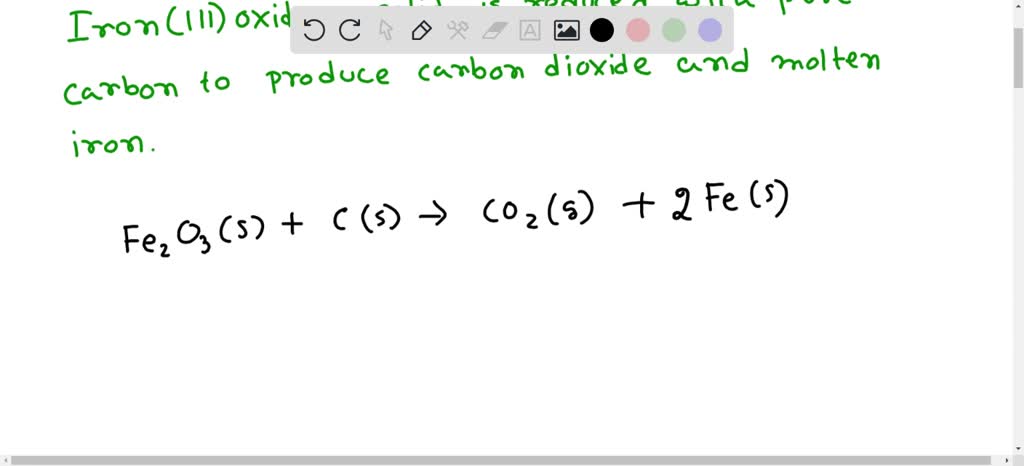
SOLVED: The balanced equation for the reaction occurring when iron(iii) oxide, a solid, is reduced with pure carbon to produce carbon dioxide and molten iron is
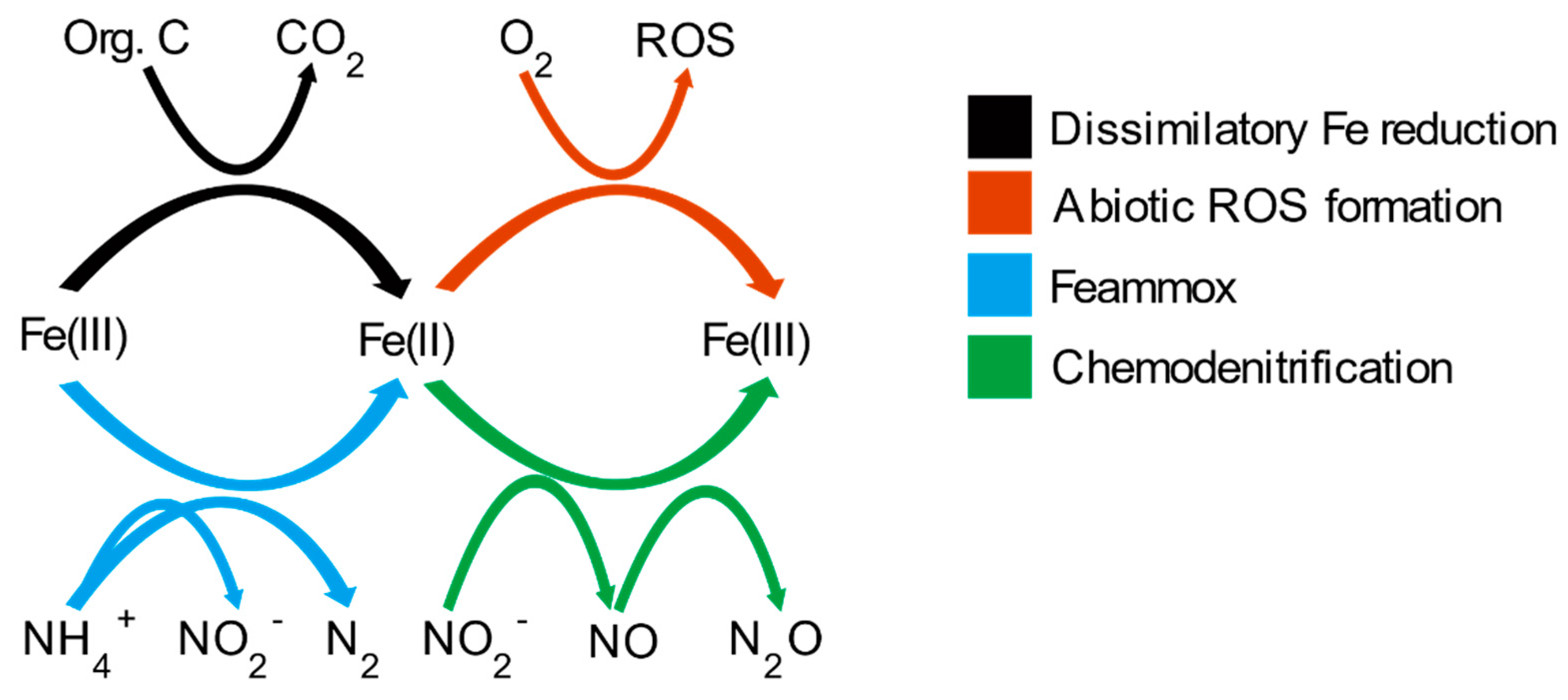
Soil Systems | Free Full-Text | Iron Redox Reactions Can Drive Microtopographic Variation in Upland Soil Carbon Dioxide and Nitrous Oxide Emissions
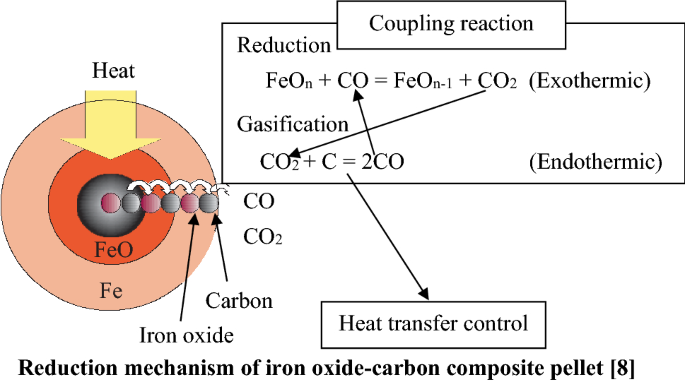
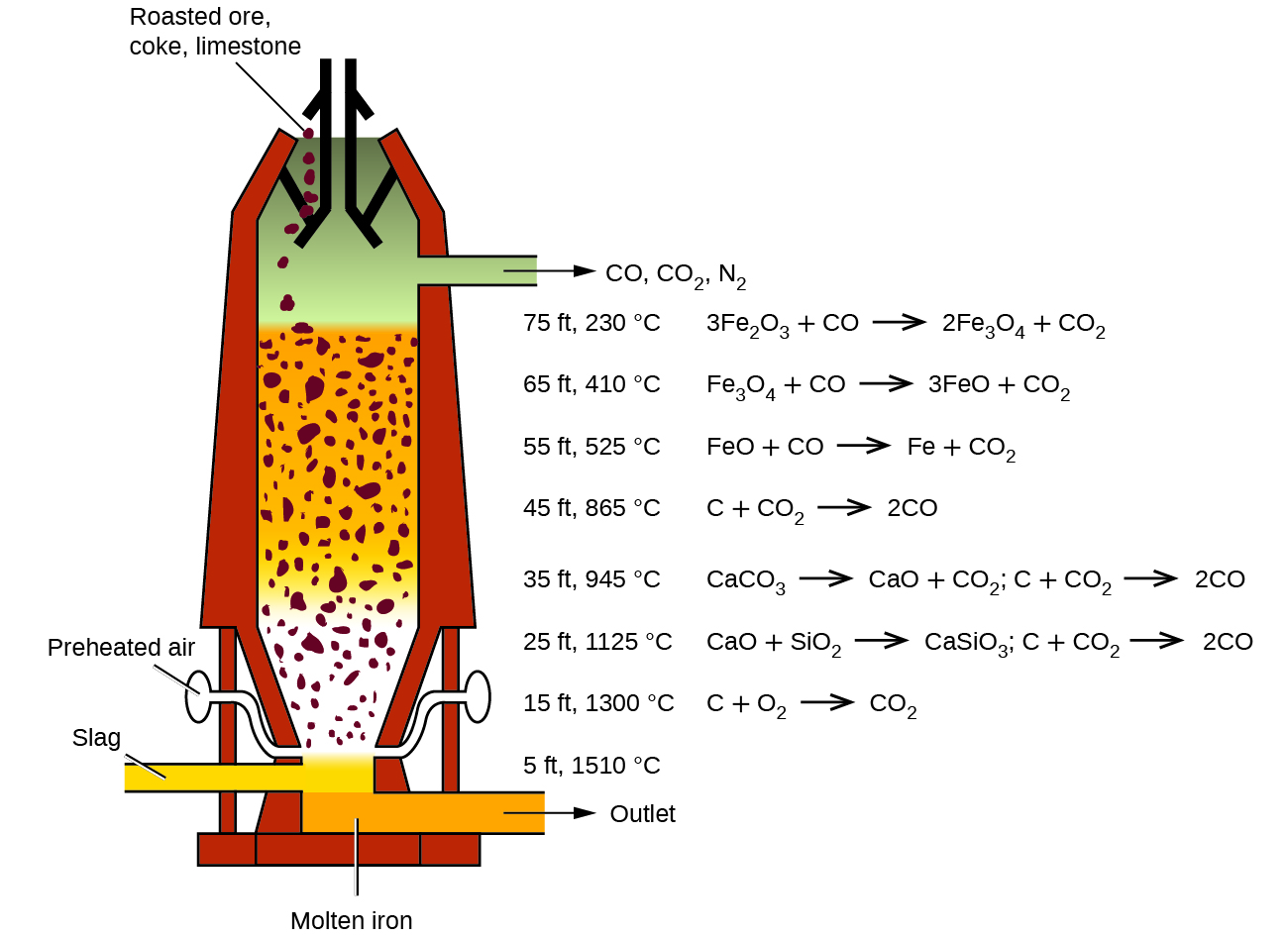
Review on Reduction Kinetics of Iron Ore–Coal Composite Pellet in Alternative and Sustainable Ironmaking | SpringerLink

Iron(iii)oxide+Carbon monoxide=Iron+ Carbon dioxide Balanced Equation ||Fe2O3+CO=Fe+CO2 Balanced Equ. - YouTube