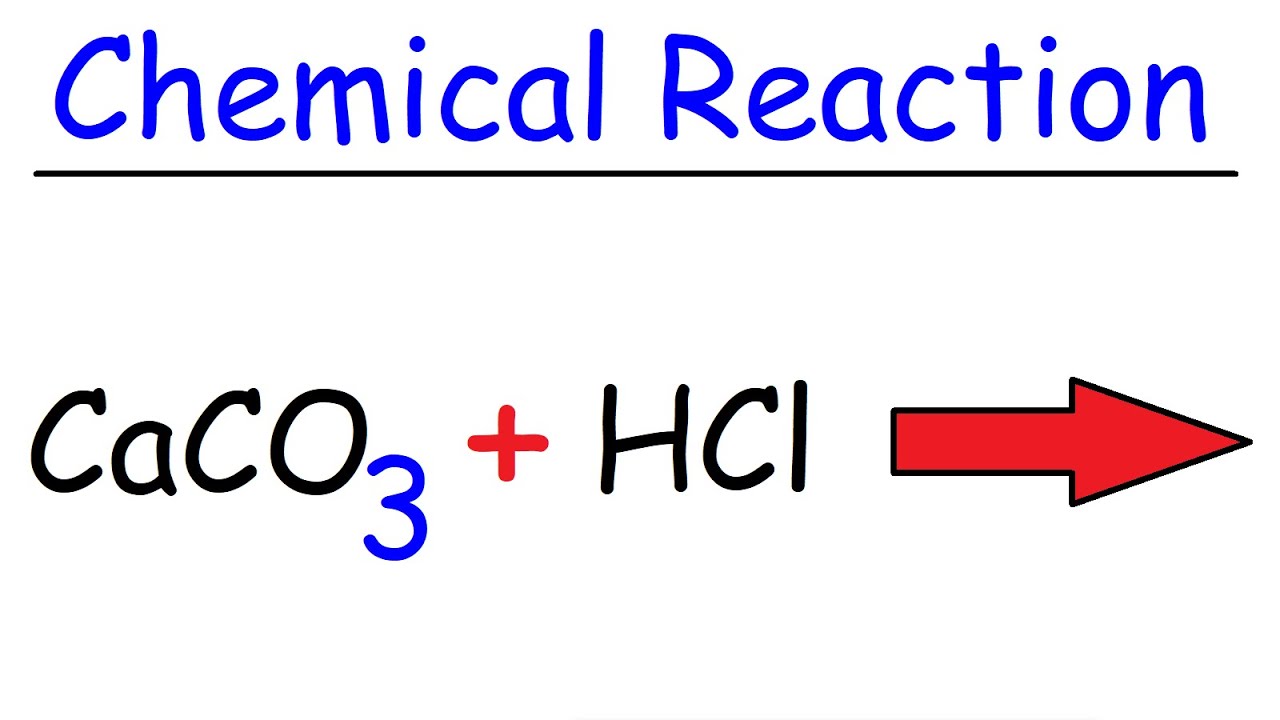
Calcium carbonate reacts with aqueous HCl to give CaCl2 and CO2 according to the reaction, CaCO3 (s) + 2HCl (aq) →CaCl2 (aq) + CO2 (g) + H2O (l) .What mass of CaCO3

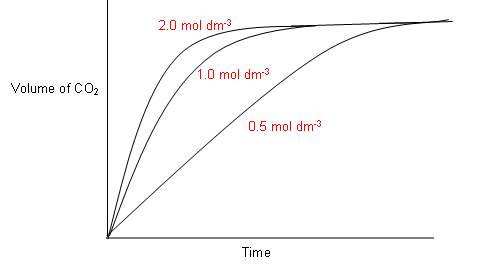
The effect of changing the concentration of hydrochloric acid on the rate of reaction with calcium carbonate - IG Exams

Chemical reactions of two calcium salts (citrate and carbonate) with... | Download Scientific Diagram

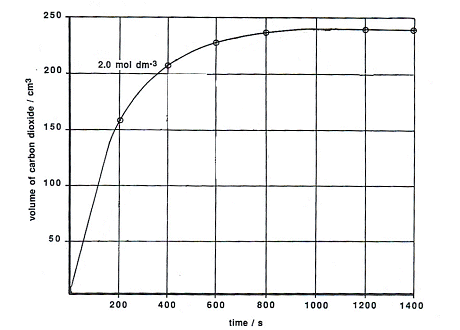
P A student carried out an experiment to investigate the rate of reaction between powdered calcium carbonate and excess dilute hydrochloric acid, using the following apparatus. The student measured the mass of

Calcium carbonate reacts with aqueous `HCl` to give `CaCl_(2)` and `CO_(2)` according to the rea... - YouTube

calcium carbonate reacts with dilute hydrochloric acid to produce carbon dioxide Stock Photo - Alamy

calcium carbonate reacts with dilute hydrochloric acid to produce carbon dioxide Stock Photo - Alamy