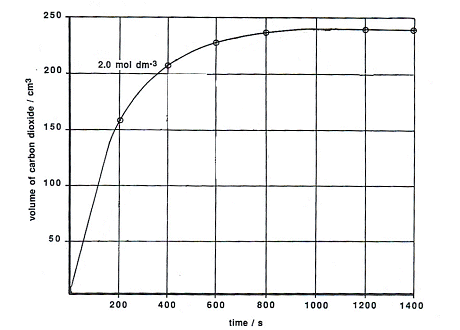
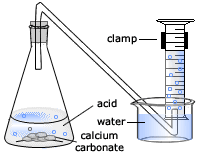
P A student carried out an experiment to investigate the rate of reaction between powdered calcium carbonate and excess dilute hydrochloric acid, using the following apparatus. The student measured the mass of

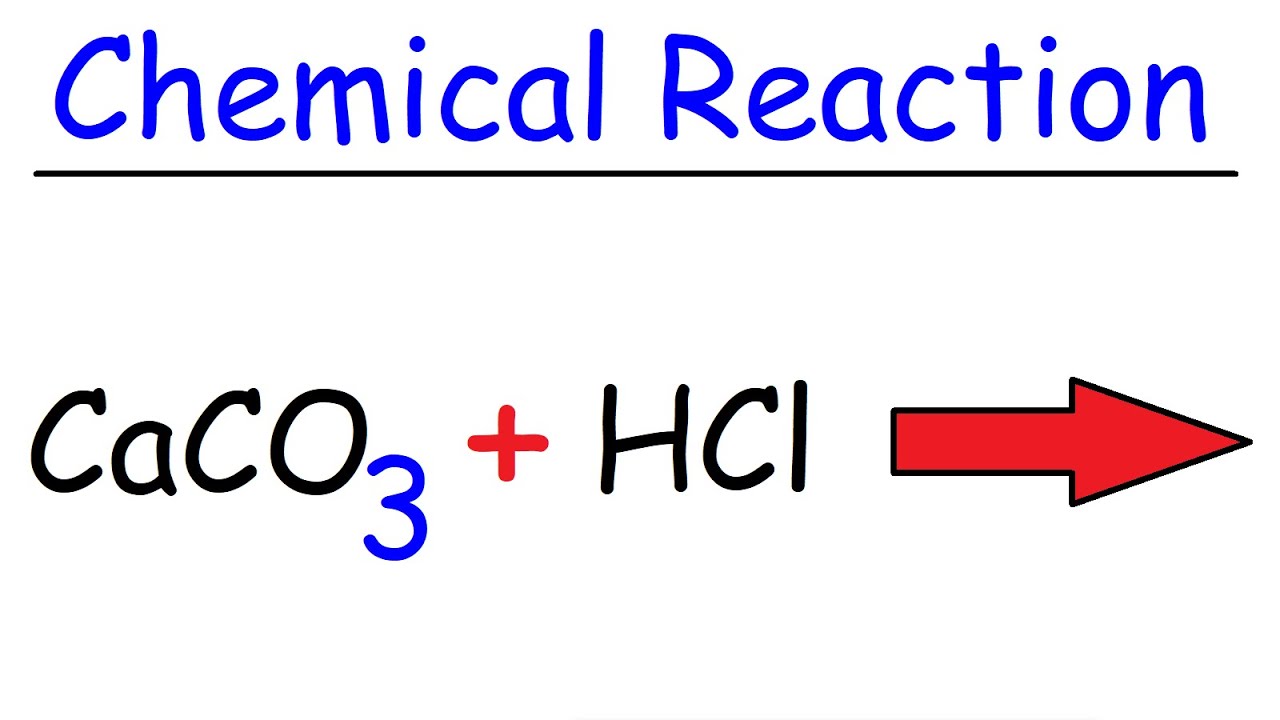
Write a balanced chemical equation for the reaction of calcium carbonate and dil. hydrochloric acid.

The effect of changing the concentration of hydrochloric acid on the rate of reaction with calcium carbonate - IG Exams

SOLVED: Queson 2 Calcium carbonate reacts with hydrochloric acid CaClz(s) according CO2(g) to the following equation: HzOn) CaCO3(s) HClg) Which of the following represents the equilibrium constant expression for the above reaction? [

Question Video: Calculating the Average Rate of Reaction of Hydrochloric Acid with Calcium Carbonate | Nagwa

Calcium carbonate reacts with aqueous HCl to give CaCl2 and CO2 according to the reaction, CaCO3 (s) + 2HCl (aq) →CaCl2 (aq) + CO2 (g) + H2O (l) .What mass of CaCO3

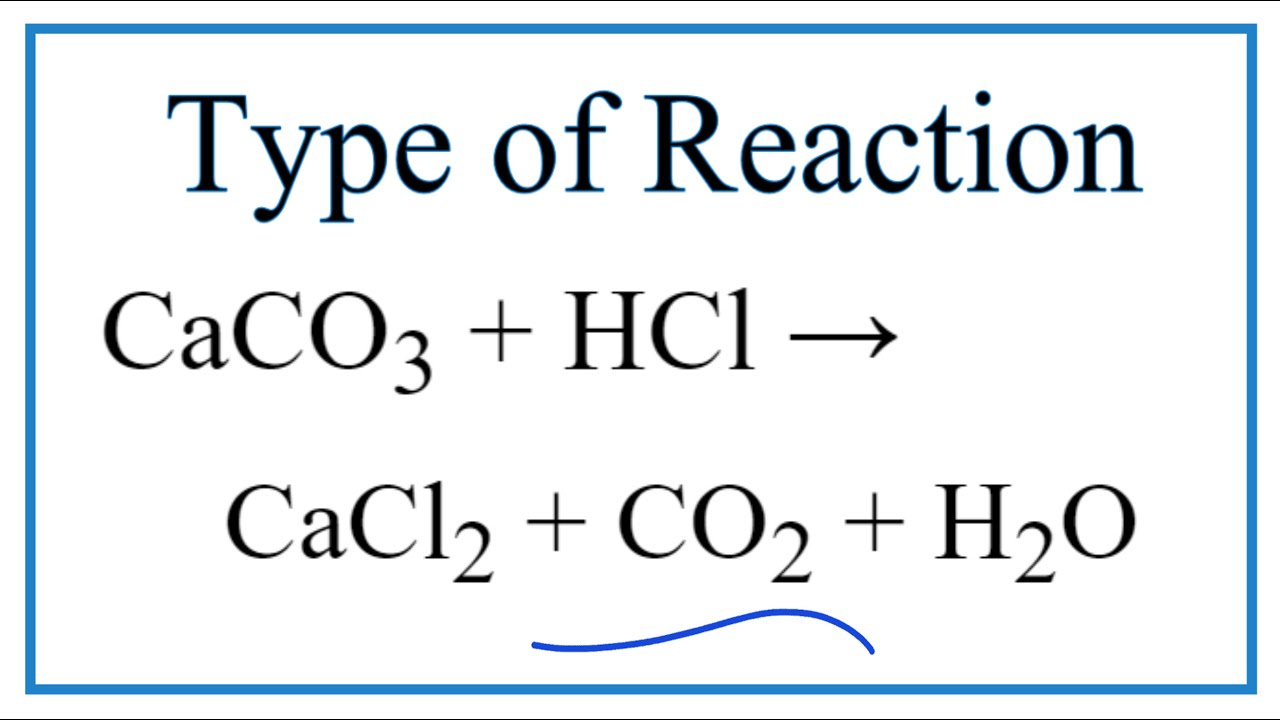
How to Balance CaCO3 + HCl = CaCl2 + CO2 + H2O | How to Balance CaCO3 + HCl = CaCl2 + CO2 + H2O Hey there! Are you struggling with balancing

calcium carbonate reacts with HCl to give CaCl2 and CO2 according to reaction.what mass of 20% impure CaCO3 - Brainly.in