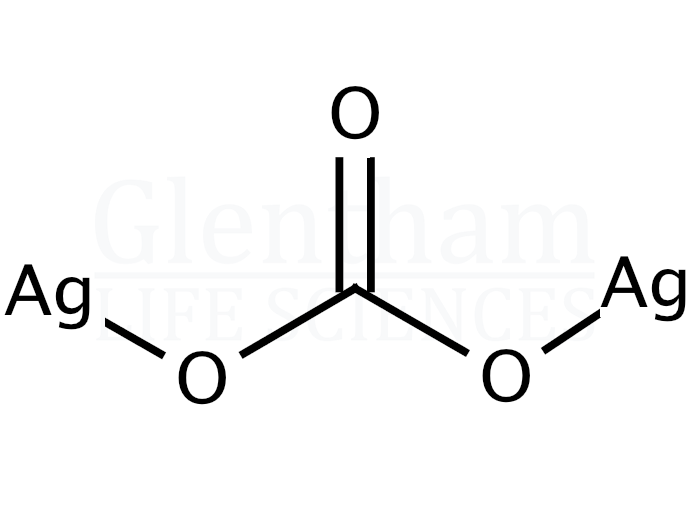
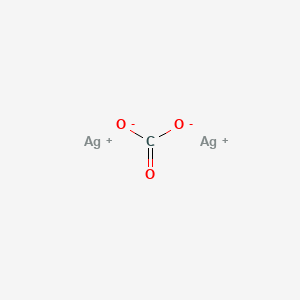
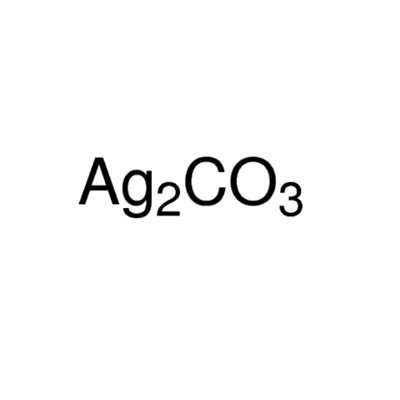Catalysts | Free Full-Text | Recent Organic Transformations with Silver Carbonate as a Key External Base and Oxidant

Q 37 2 76g of silver carbonate on being strongly heated yield a residue weighing 37 2 76 - Chemistry - Some Basic Concepts of Chemistry - 12831329 | Meritnation.com
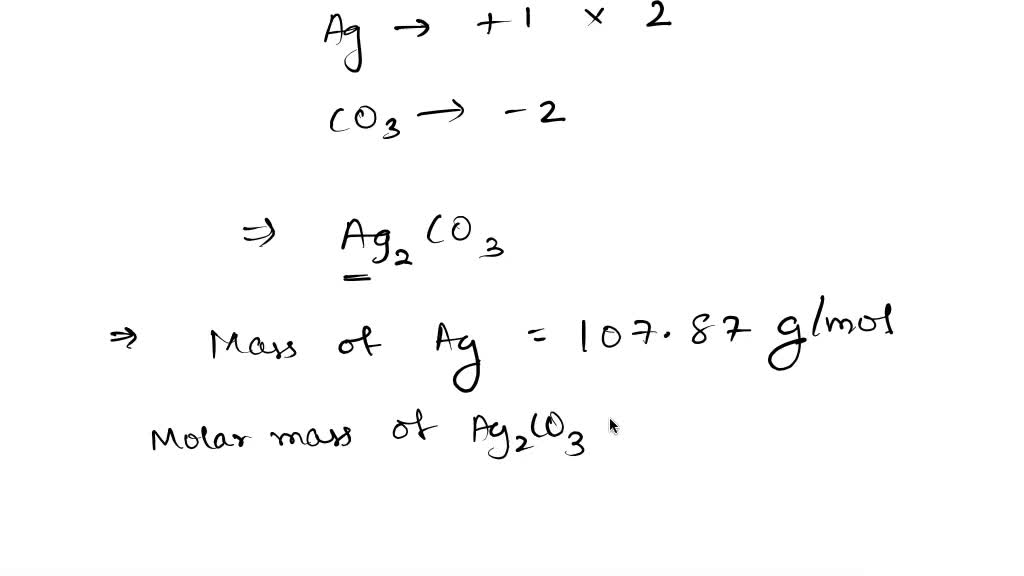
SOLVED: Calculate the percent by mass of silver found in a particular mineral that is determined to be silver carbonate.
![SOLVED: Cosider the dissociation of silver carbonate: Ag2COz (s) == 2Ag+ (aq) + CO32- (aq) (Ksp = 8.10 . 10-13 When the molarities of ions are: [Agt] =2.30 10-4M, and [CO32-] = SOLVED: Cosider the dissociation of silver carbonate: Ag2COz (s) == 2Ag+ (aq) + CO32- (aq) (Ksp = 8.10 . 10-13 When the molarities of ions are: [Agt] =2.30 10-4M, and [CO32-] =](https://cdn.numerade.com/ask_images/aa22dd22039a420ea2e6f173a3cd9160.jpg)