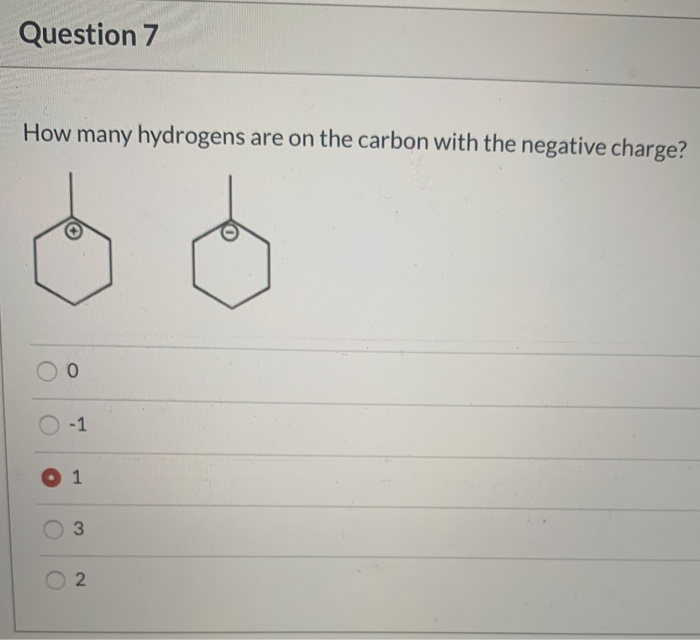
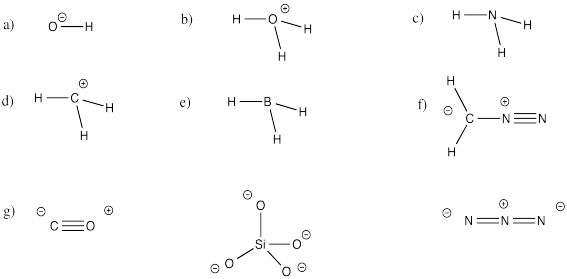
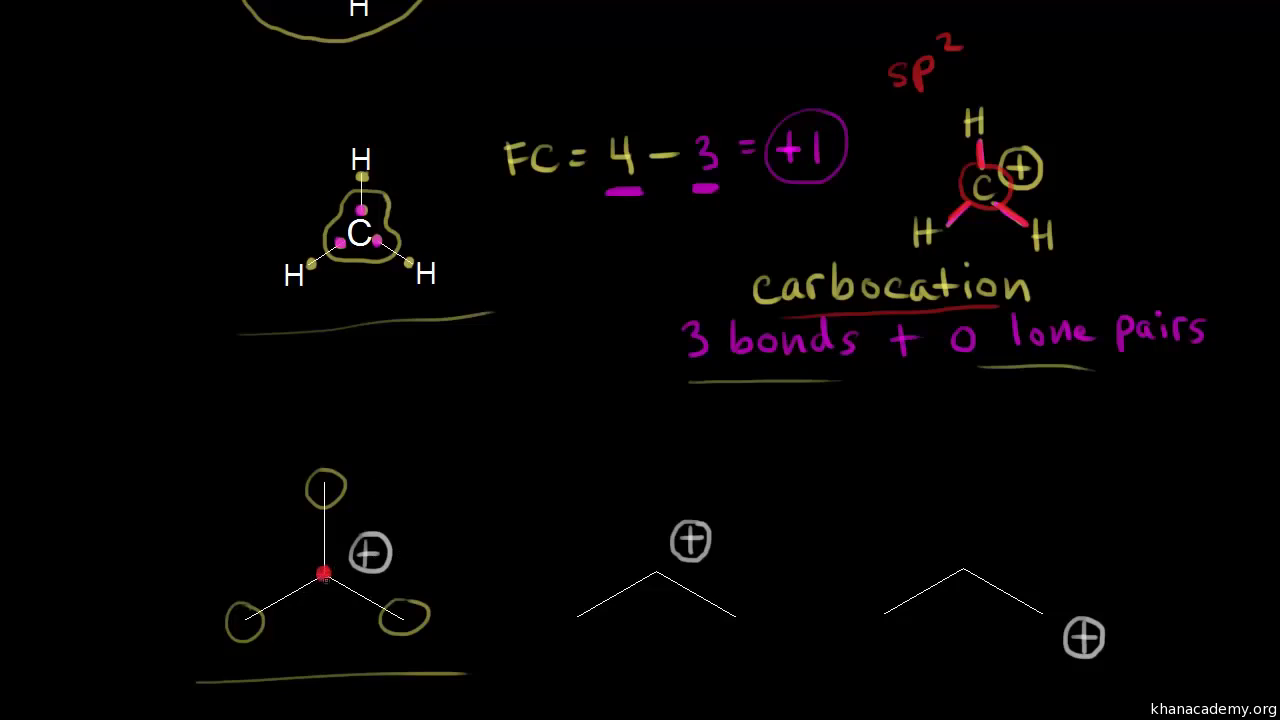
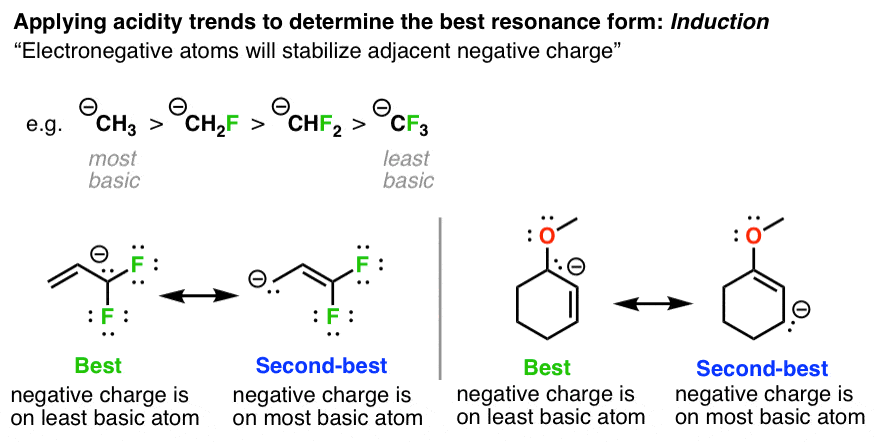
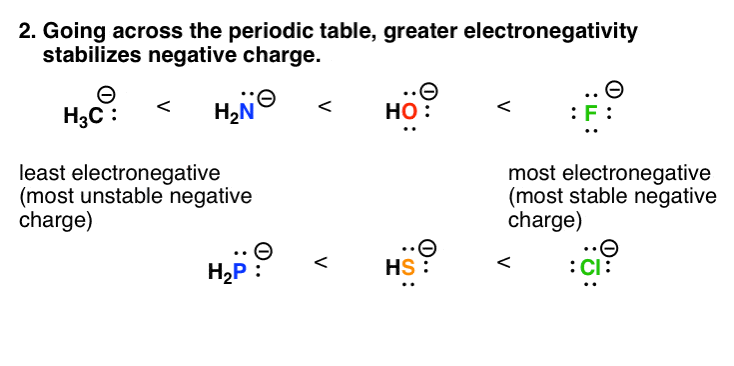
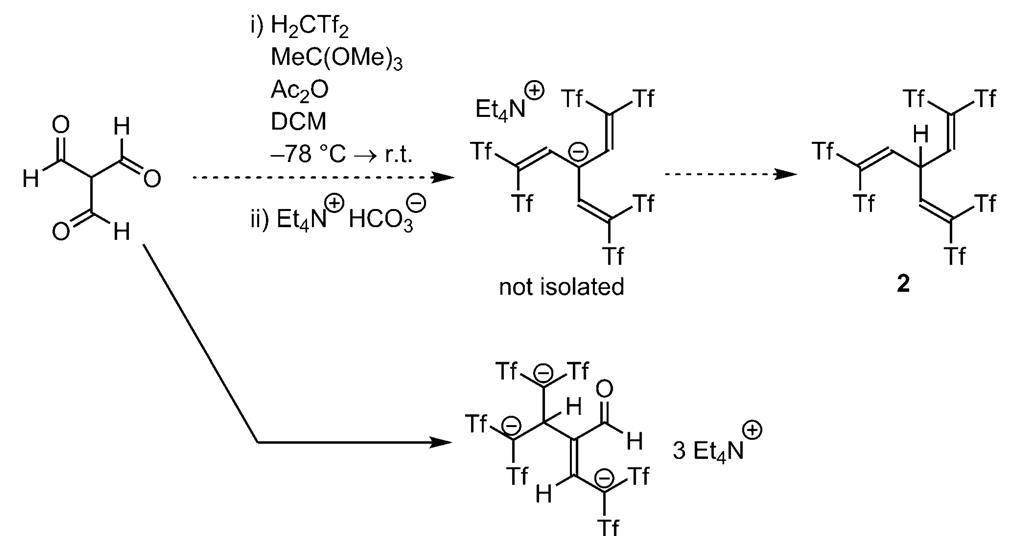
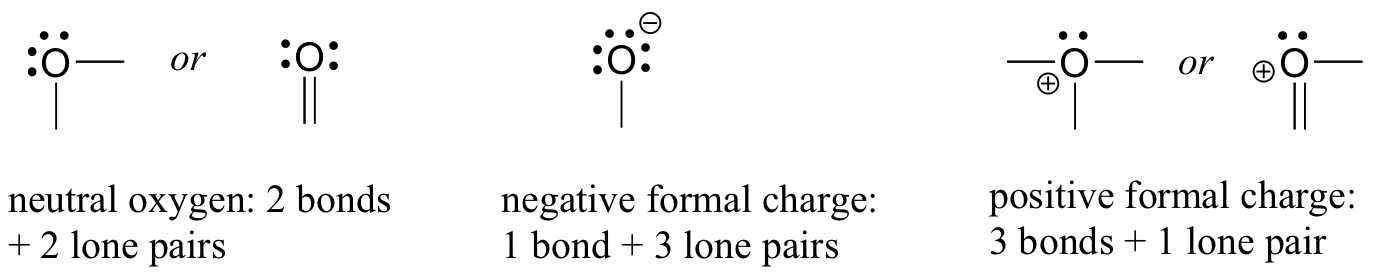
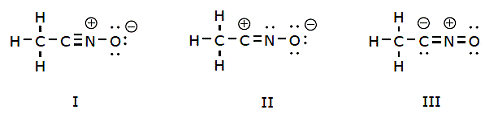Classification of Negative Charge Discriminate Hybridization with Aromatic and Anti-aromatic Behavior of Organic Compounds - Innovative Mnemonics

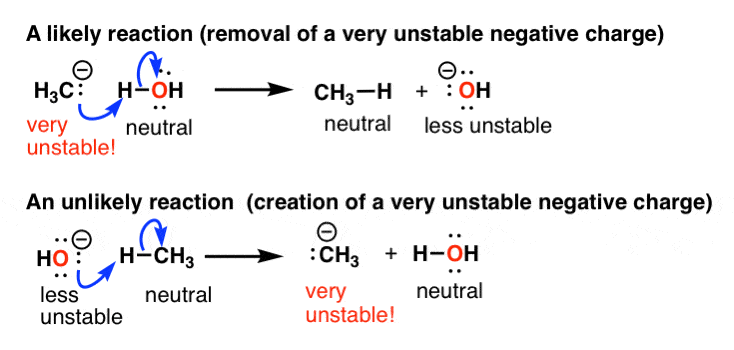
Why the Wittig reagent exist as a negative charge on carbon and a positive charge on phosphorus? My first thought was due to electro negativity but it doesn't seem right as the

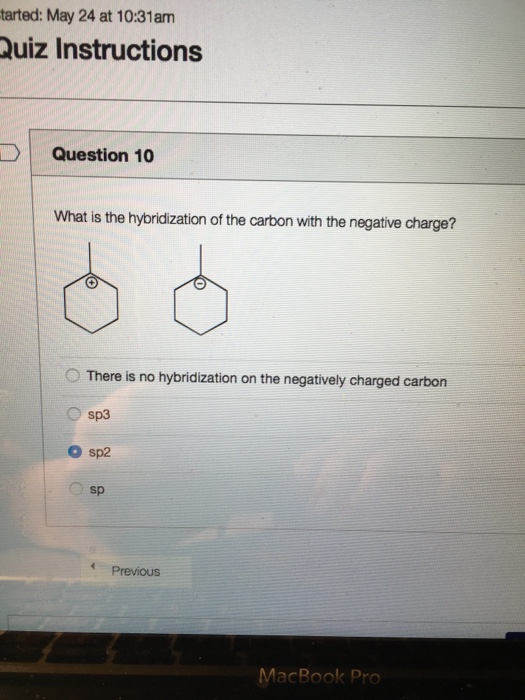
Hybridisation of carbon bearing negative charge inthe following structure is(1) sp(2) sp2(4) sp3d(3) sp3 - Brainly.in

If a negative charge is created on carbon in a cyclic ring, does the hybridisation status of the carbon change, and if this does happen, why? - Quora
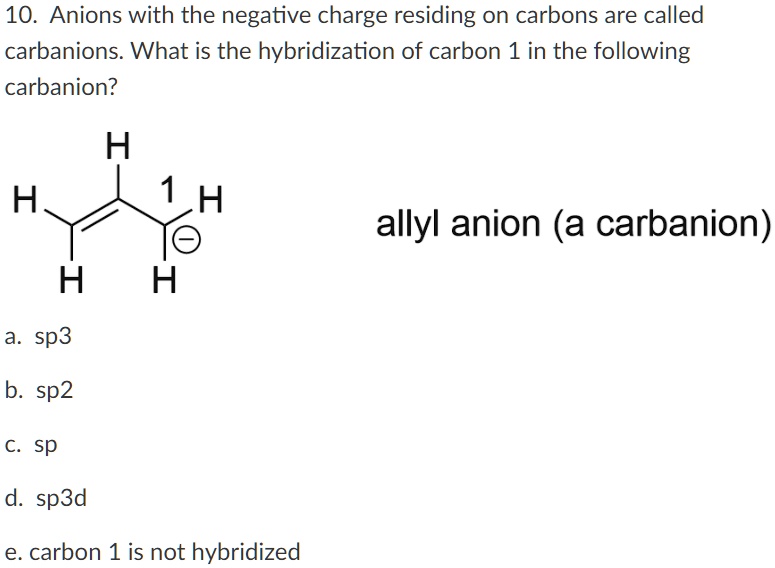
SOLVED: 10. Anions with the negative charge residing on carbons are called carbanions What is the hybridization of carbon 1 in the following carbanion? H 1 H O H allyl anion (a

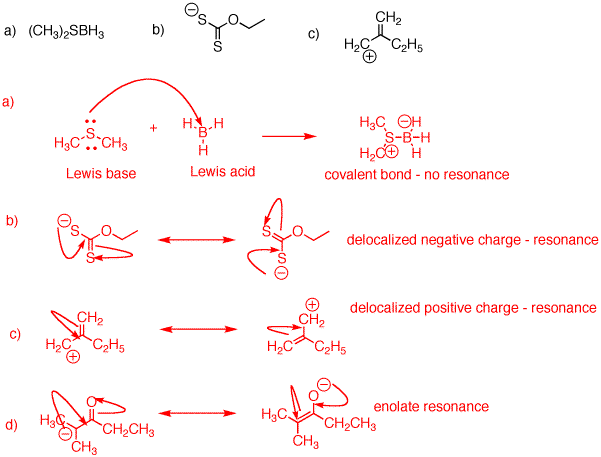
organic chemistry - Shouldn't the negative charge be on carbon in this scenario according to molecular orbital theory - Chemistry Stack Exchange