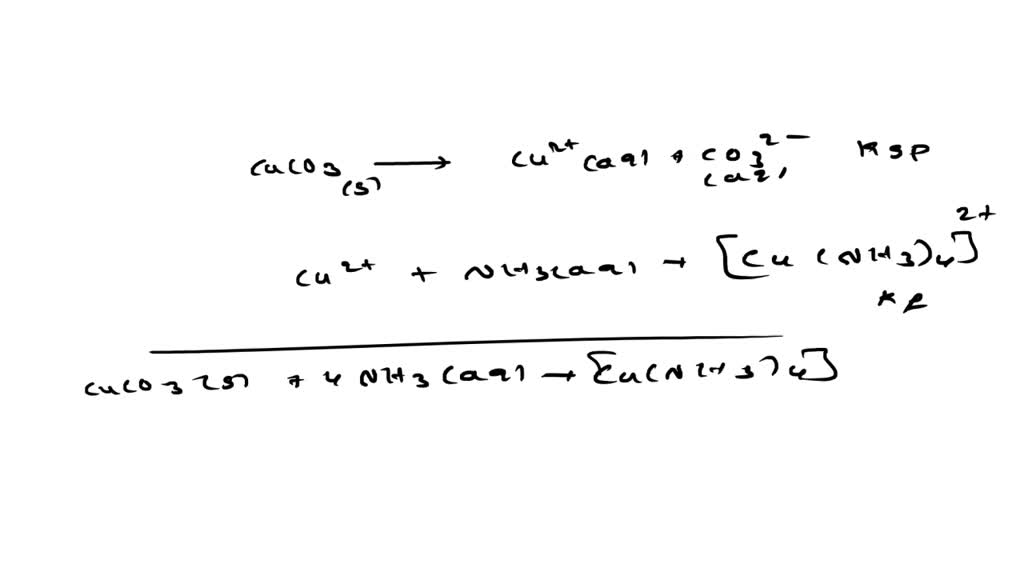
SOLVED: Copper carbonate is not very soluble in water, but it will dissolve in ammonia solutions because it forms a complex ion. Given the equilibrium constants below, what is the solubility of

gcse Formulae oxides, hydroxides, carbonates, chlorides, sulphates/sulfates, nitrates, salts solubility curves & water of crystallisation data tables KS3 KS4 Science IGCSE chemistry revision notes



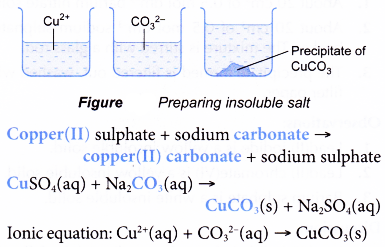
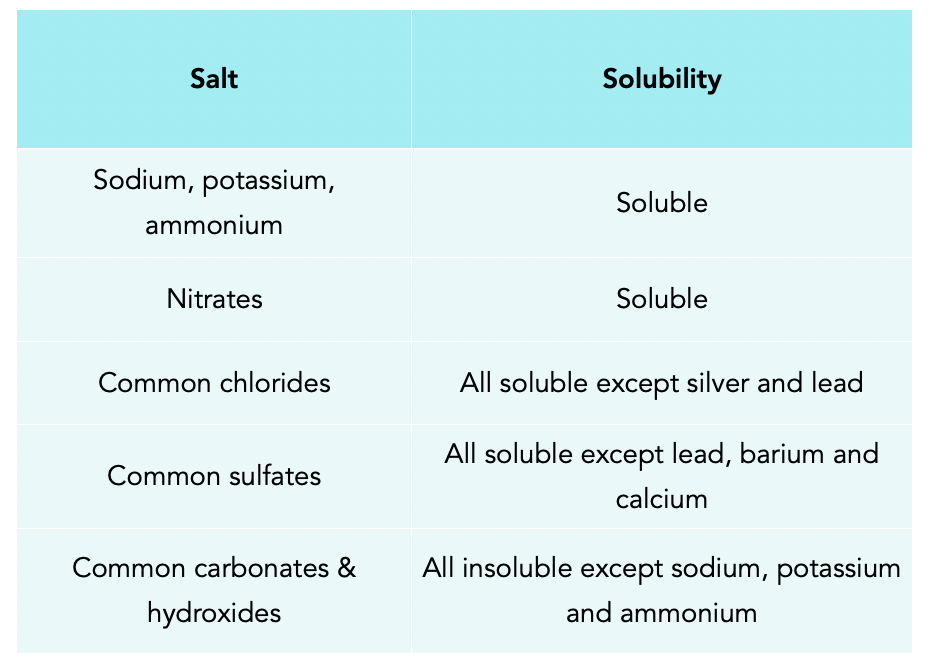
![Solubility of Salts [Online Video] – O Level Secondary Chemistry Tuition Solubility of Salts [Online Video] – O Level Secondary Chemistry Tuition](https://icandochemistry942105908.files.wordpress.com/2019/02/salt-solubility.jpg?w=1200)










