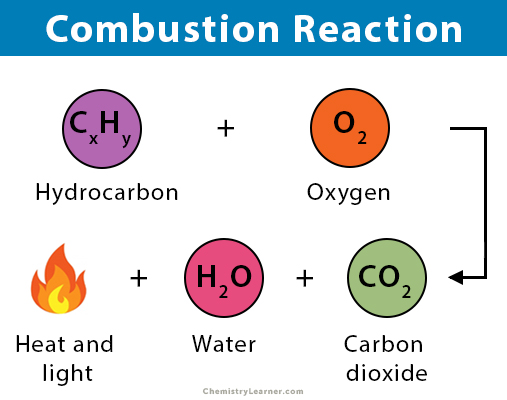
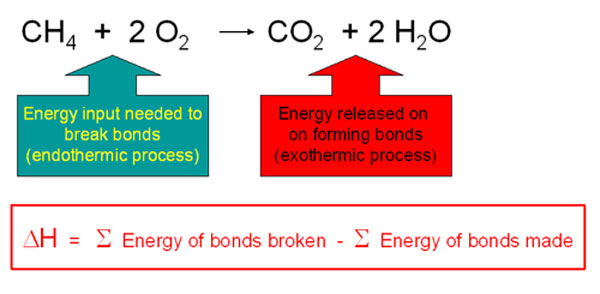
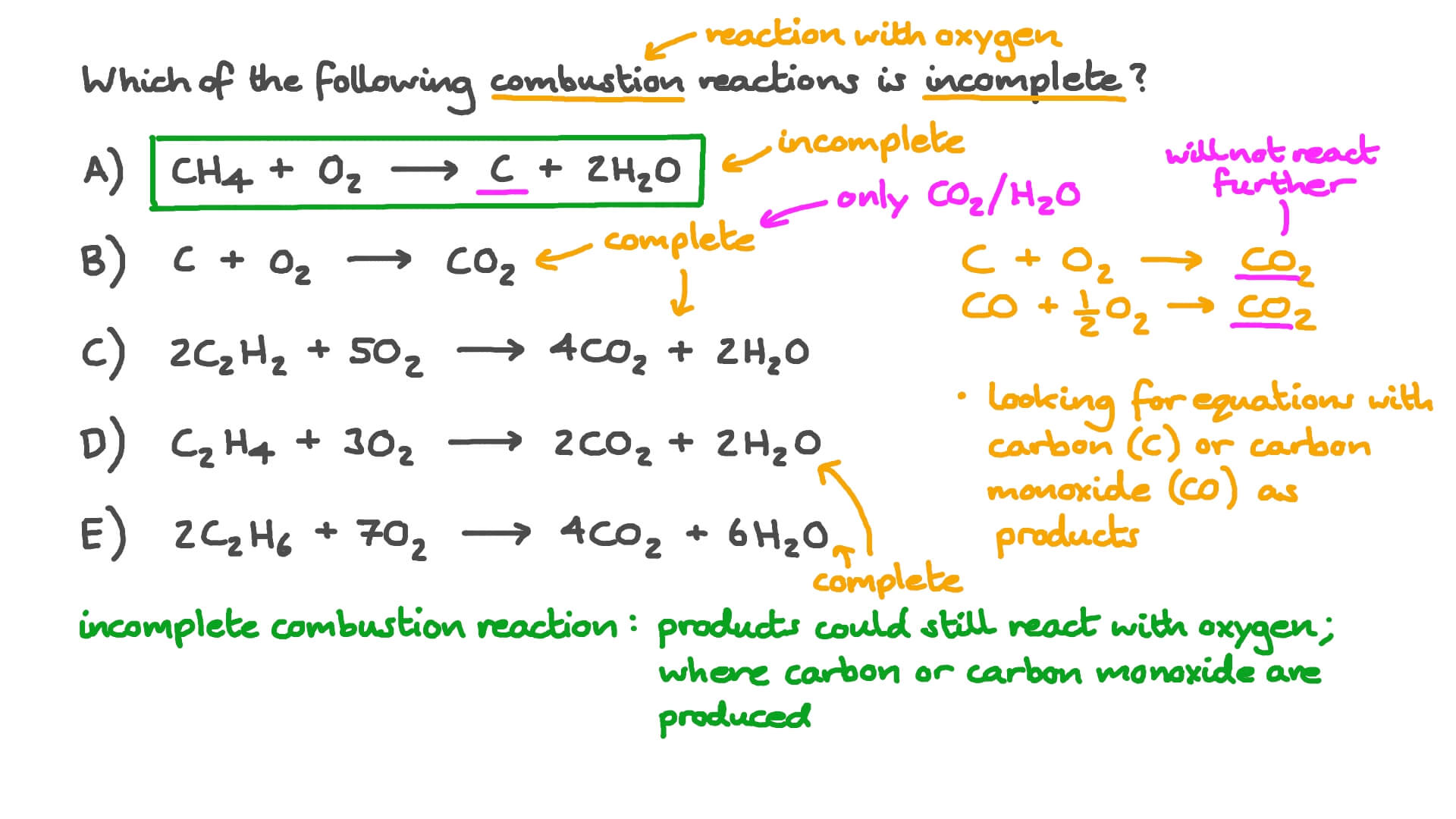
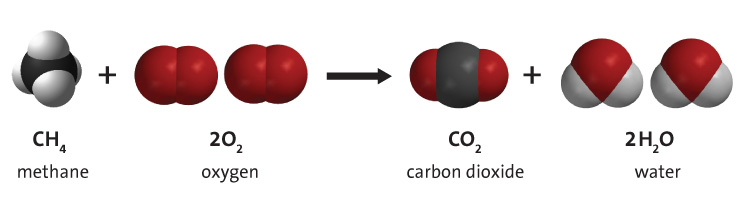
Write the balanced chemical equation for combustion of carbon dioxide - Science - Chemical Reactions and Equations - 12555075 | Meritnation.com

CHE Combustion Reactions Fuels –Gaseous –Liquid –Solid Oxidants Elementary combustion reactions Theoretical and excess air. - ppt download

SOLVED: The incomplete combustion of carbon is described by the following equation: 2C () + Oz (g) 2C0 (2) heat energy For this reaction, write the appropriate AH, 4S and AG on
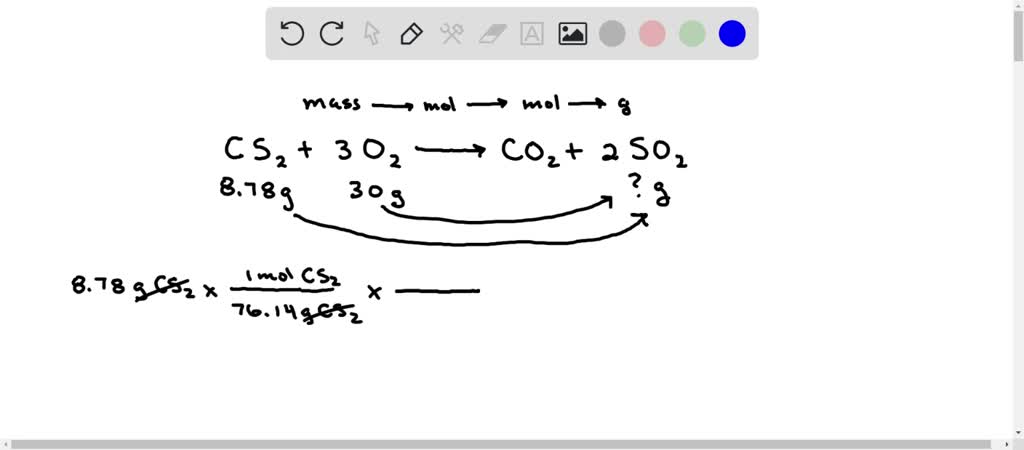
SOLVED: The combustion of carbon disulfide in the presence of excess oxygen yields carbon dioxide and sulfur dioxide according to the following UNBALANCED reaction: CS2 (g) + 3O2 (g) → CO2 (g) +
20. One mole of carbonundergoes incomplete combustion to produce carbon monoxide. Calculate(AH Δ ) for the formation of CO at 298 KGiven R 8314 JK 1 mol 1
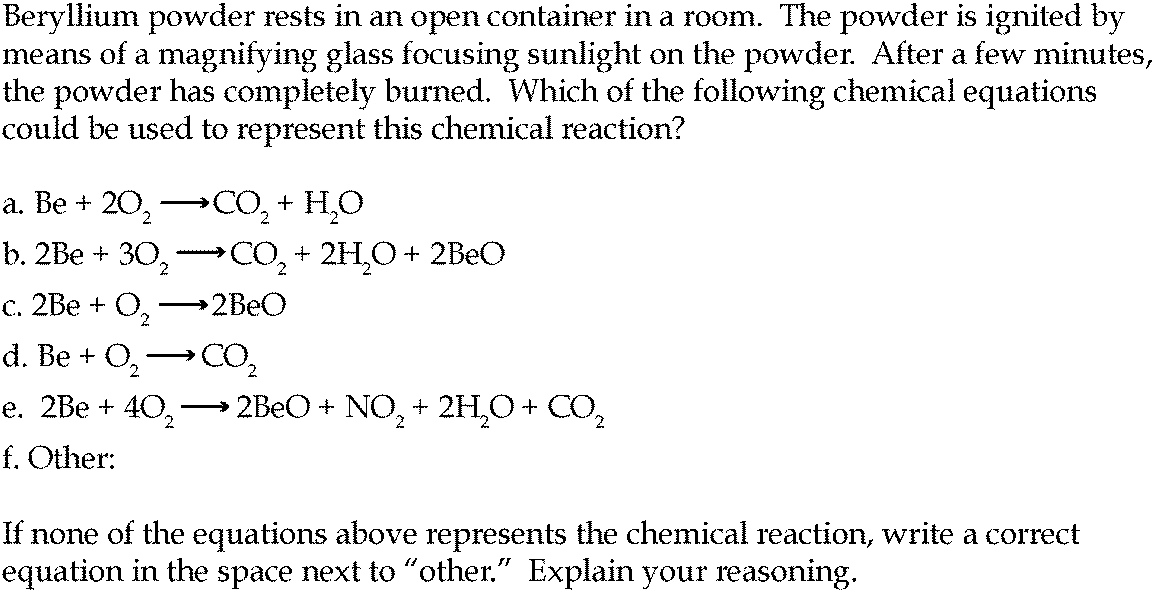
Combustion always produces carbon dioxide and water”: a discussion of university chemistry students' use of rules in place of principles - Chemistry Education Research and Practice (RSC Publishing) DOI:10.1039/C4RP00089G