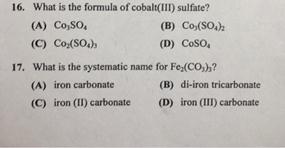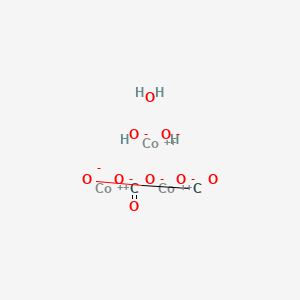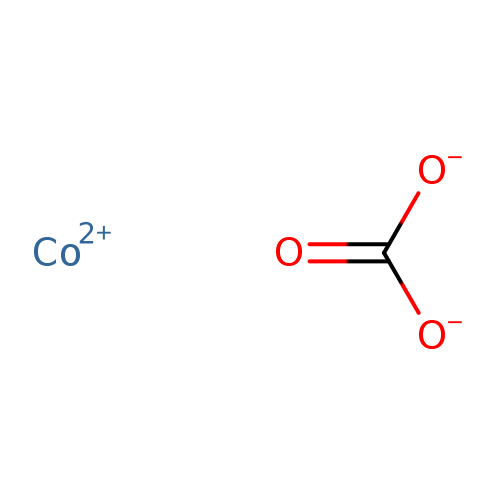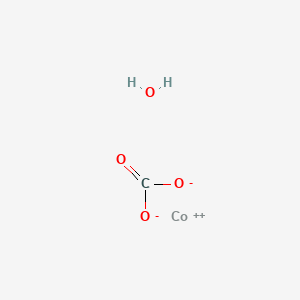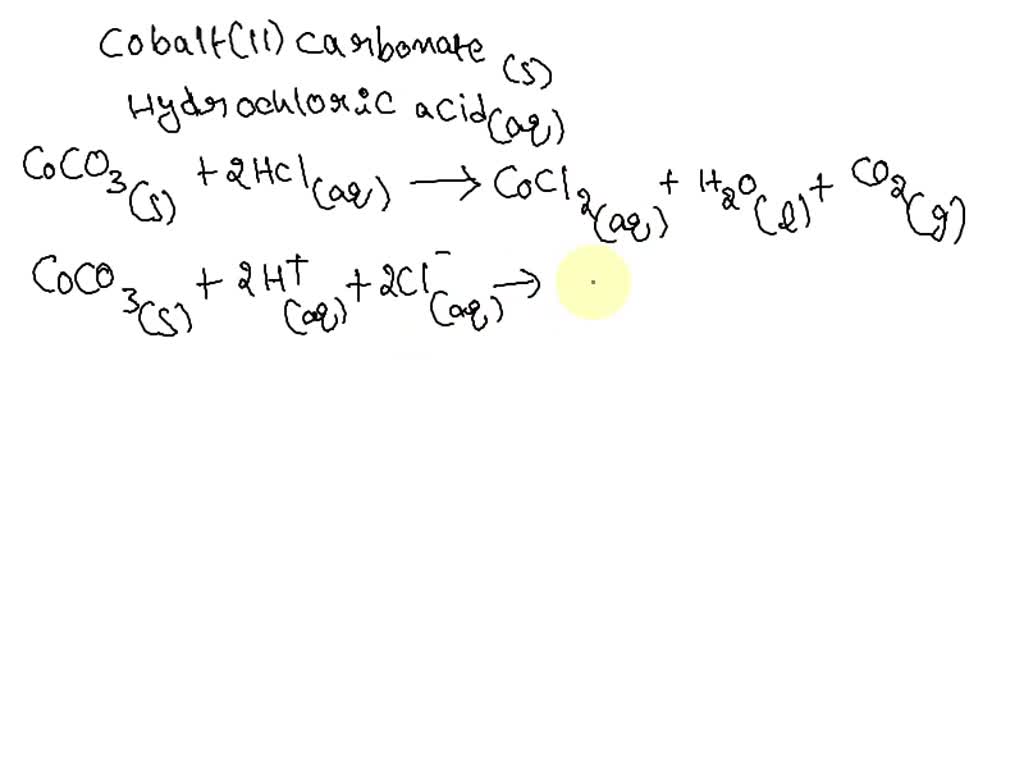
SOLVED: Write a net ionic equation for the reaction that occurs when cobalt( II) carbonate (s) and excess hydrochloric acid (aq) are combined.
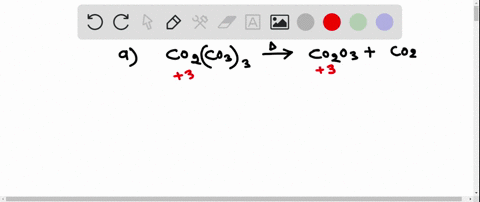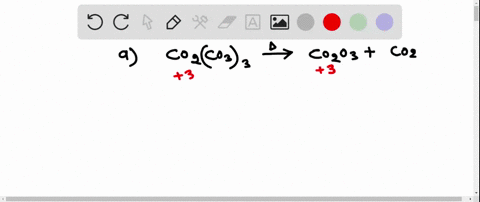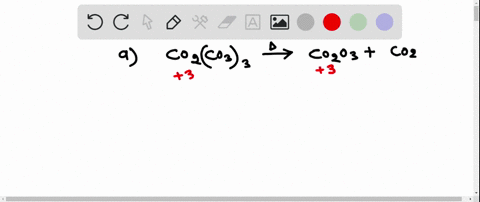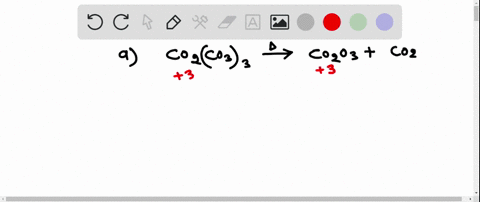
SOLVED:Write a balanced equation for each of the following decomposition reactions: (a) Cobalt(III) carbonate decomposes with heat to give solid cobalt(III) oxide, and carbon dioxide gas. (b) Tin(IV) carbonate decomposes with heat

SOLVED:Write a balanced equation for each of the following decomposition reactions: (a) Cobalt(III) carbonate decomposes with heat to give solid cobalt(III) oxide, and carbon dioxide gas. (b) Tin(IV) carbonate decomposes with heat
![SOLVED: The net ionic equation for the reaction of a cobalt(II) sulfate solution and a sodium carbonate solution is: [a] Co2+ (aq) + CO3 2̶(aq) + 2 Na+ (aq) + SO4 2- ( SOLVED: The net ionic equation for the reaction of a cobalt(II) sulfate solution and a sodium carbonate solution is: [a] Co2+ (aq) + CO3 2̶(aq) + 2 Na+ (aq) + SO4 2- (](https://cdn.numerade.com/ask_previews/d76bf241-7907-4c05-a2e2-3a50f51ffda2_large.jpg)
SOLVED: The net ionic equation for the reaction of a cobalt(II) sulfate solution and a sodium carbonate solution is: [a] Co2+ (aq) + CO3 2̶(aq) + 2 Na+ (aq) + SO4 2- (

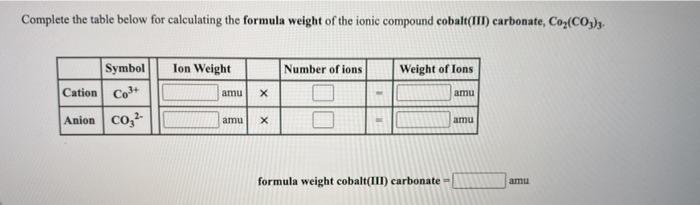
Q2 Complete the worksheet at your table. Be sure to read instructions carefully and begin working promptly. - ppt download