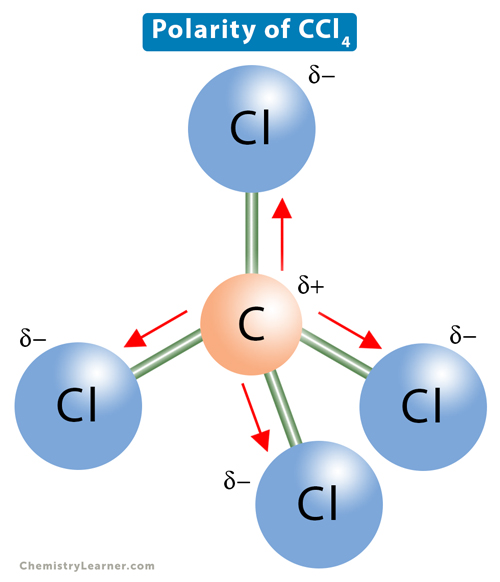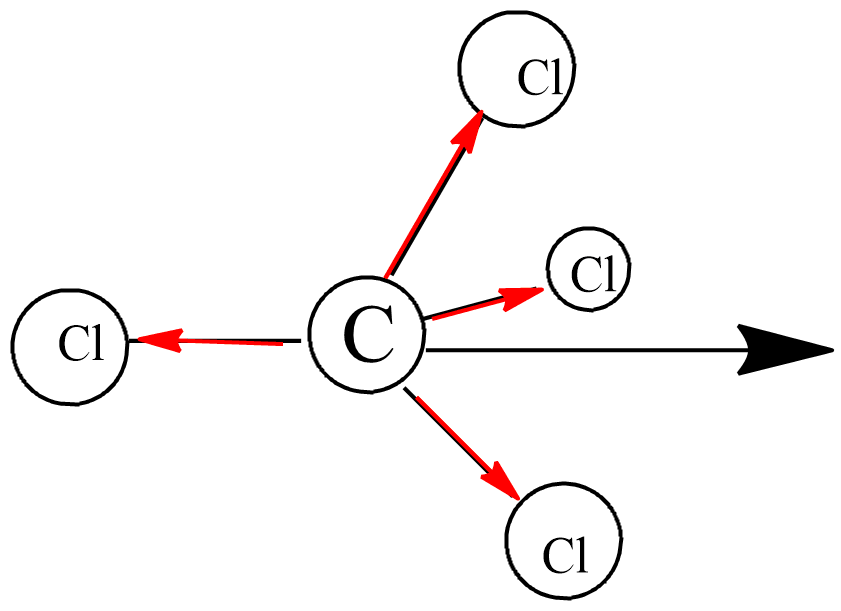
Carbon tetrachloride ($CC{{l}_{4}}$ ):(A) ionic substance(B) non-polar covalent substance(C) polar covalent substance (D) macromolecular substance(E) metallic substance
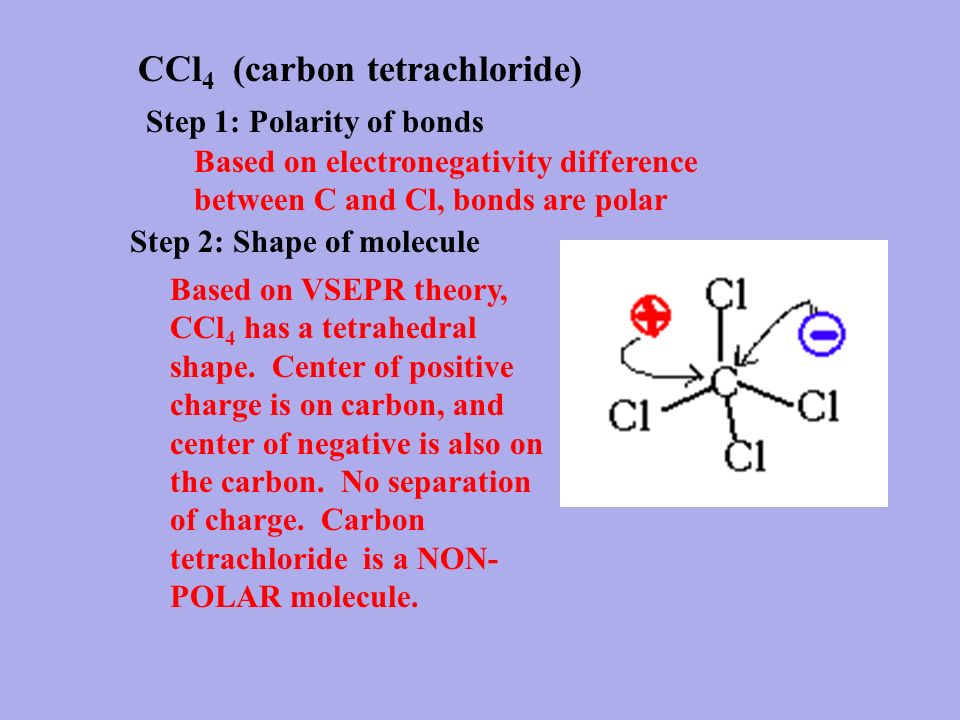
Polarity of Molecules 11/18/14 Polar Molecules are molecules which have an uneven distribution of charge. One side of the molecule is negative while. - ppt download

Draw the Lewis dot structure for CCl4. Determine the electron geometry and molecular shape of this molecule. Is this molecule polar or nonpolar? | Homework.Study.com
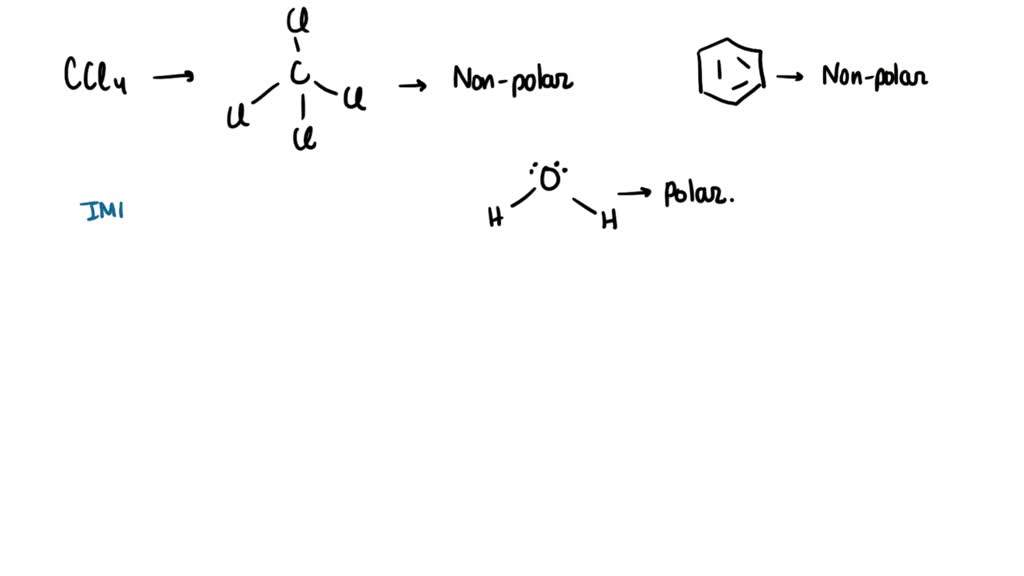
SOLVED: Carbon tetrachloride, CCI4(l), is more mixable with benzene, C6H6(l), than with H2O because (1) the intermolecular forces are similar in both carbon tetrachloride and benzene. (2) both carbon tetrachloride and benzene




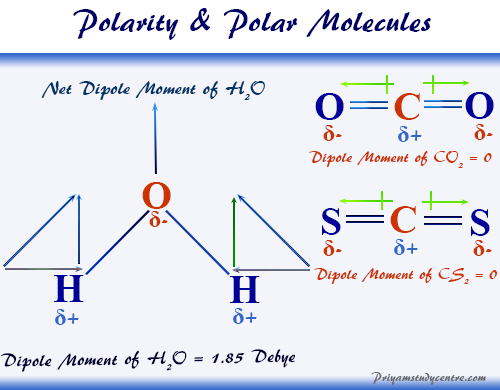
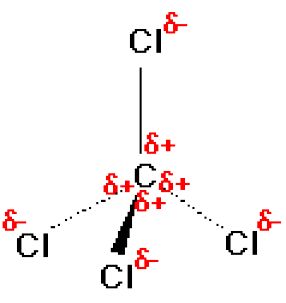








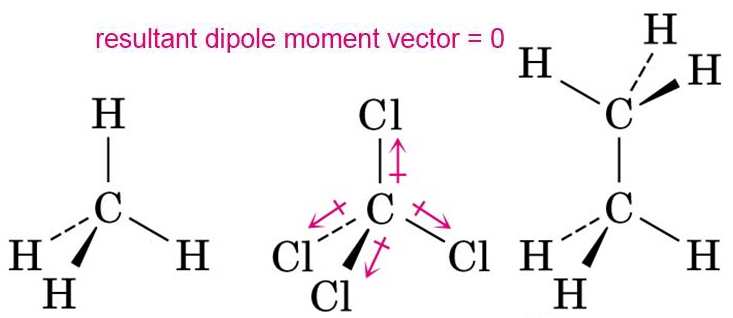

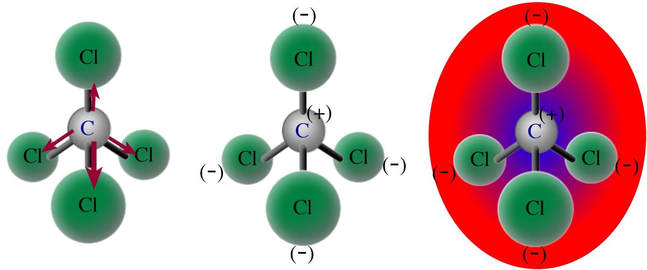

![Is \\[CC{l_4}\\] polar or nonpolar? Is \\[CC{l_4}\\] polar or nonpolar?](https://www.vedantu.com/question-sets/dbf3f5ee-35e7-43b7-b8a3-cfe63dfd520e2097468885289678636.png)