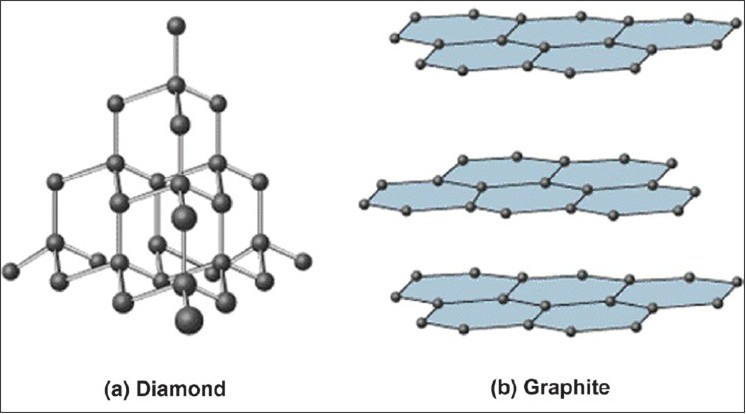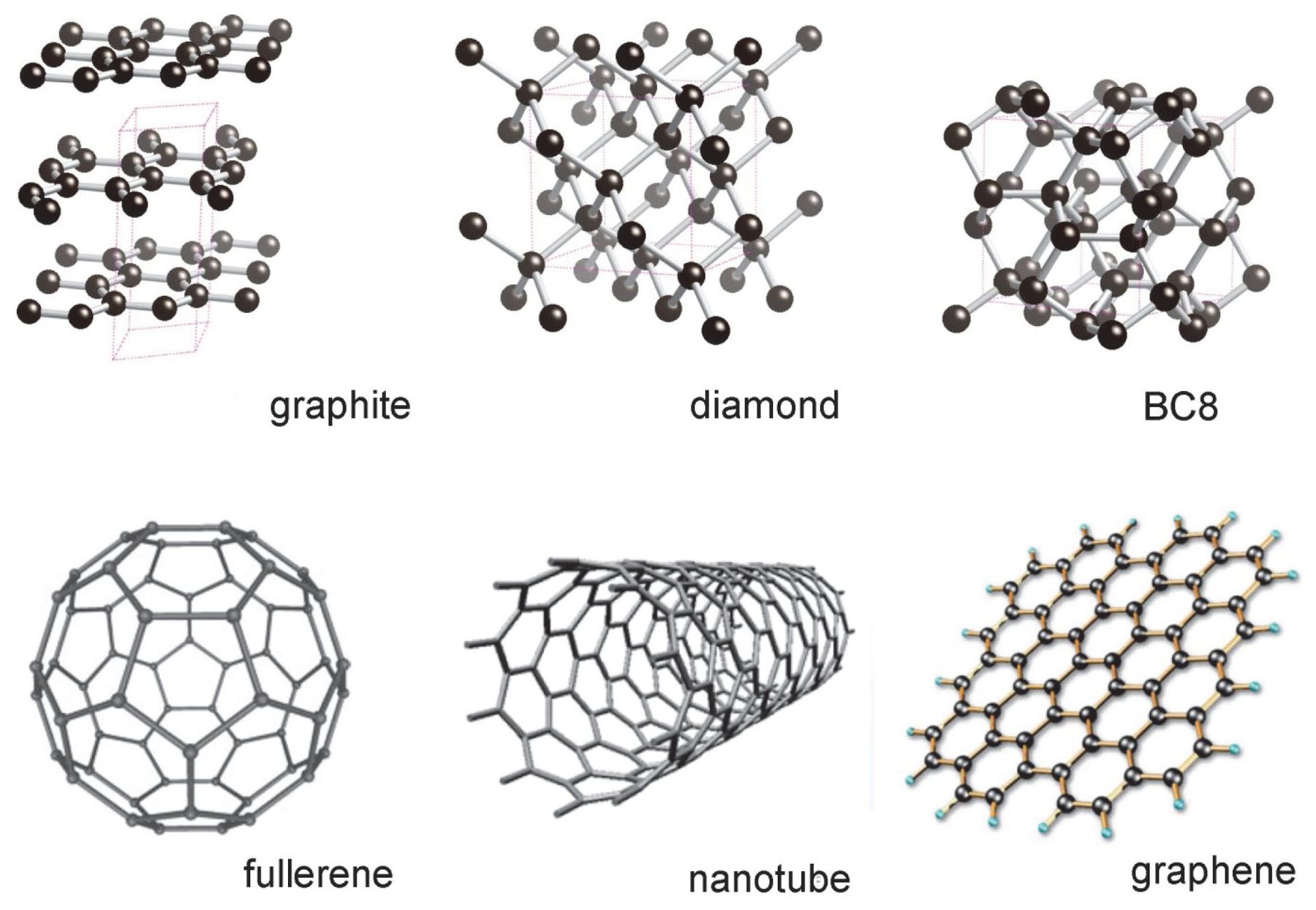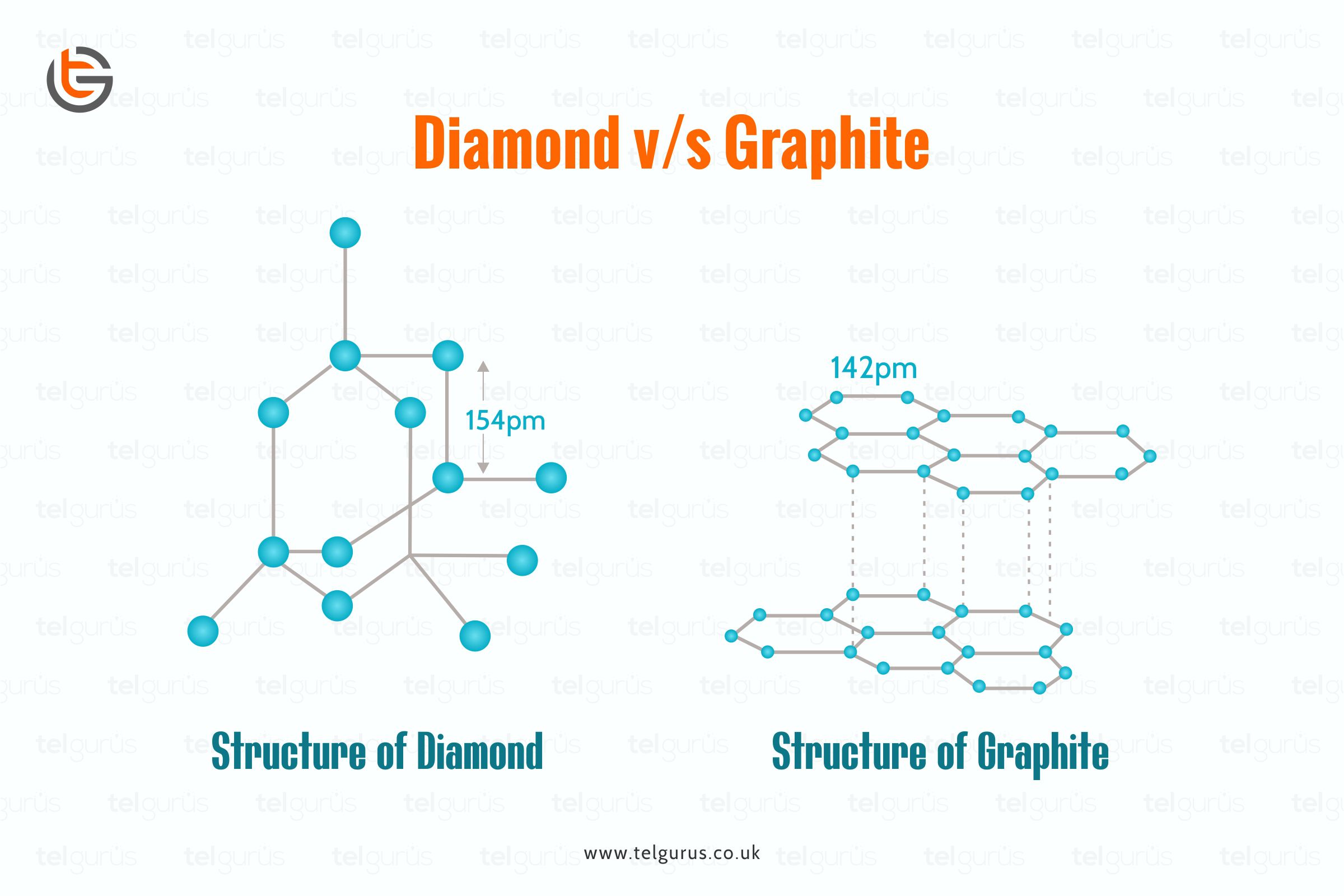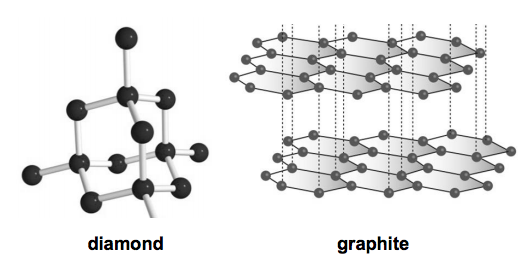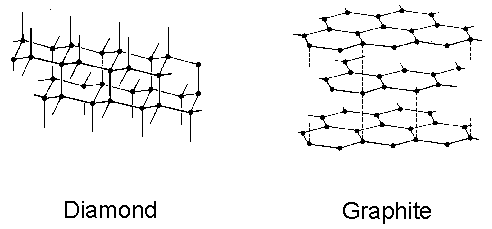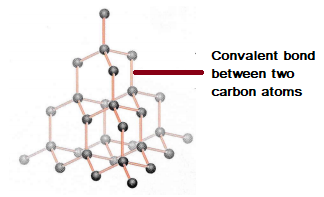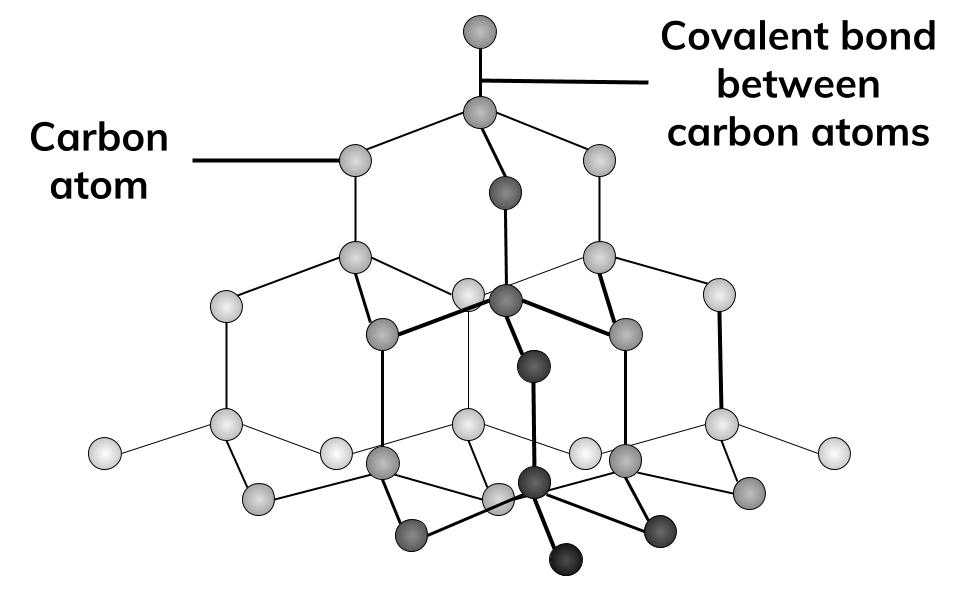
1:50 explain how the structures of diamond, graphite and C60 fullerene influence their physical properties, including electrical conductivity and hardness - TutorMyself Chemistry

Ideal and real structures of different forms of carbon, with some remarks on their geological significance | Journal of the Geological Society

UNM Foundation Engineering - Do you know that diamond and graphite are made from the same element? Source: https://www.bbc.co.uk/bitesize/guides/zspxdxs/revision/2 . . . . . . #WeAreUoN #UoNMalaysia #UNM #nottinghammalaysia #nottinghamuniversity ...

Why diamond is so shiny whereas graphite is not shiny despite the fact that both are made up of carbon? - Quora

Graphite and diamond have the same composition, but how do their properties have great difference with each other like their structure, price, color, etc.? - Quora
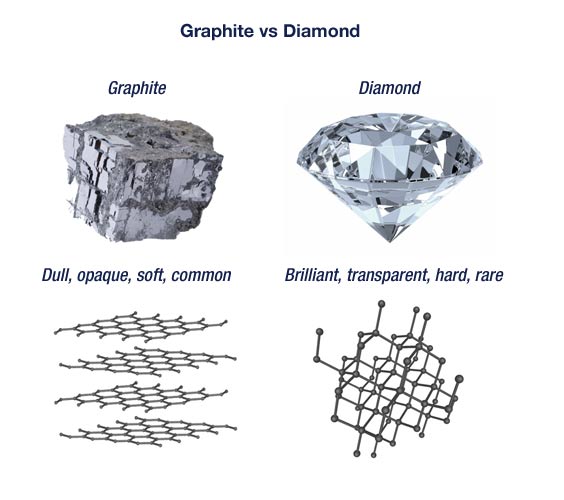
butterfly110 on Twitter: "How can graphite and diamond be so different if they are both composed of pure carbon? #Bio110Fall18 @NutmegSomething https://t.co/d70J08szHV" / Twitter