![The biologically mediated water–gas shift reaction: structure, function and biosynthesis of monofunctional [NiFe]-carbon monoxide dehydrogenases - Sustainable Energy & Fuels (RSC Publishing) The biologically mediated water–gas shift reaction: structure, function and biosynthesis of monofunctional [NiFe]-carbon monoxide dehydrogenases - Sustainable Energy & Fuels (RSC Publishing)](https://pubs.rsc.org/en/Content/Image/GA/C8SE00085A)
The biologically mediated water–gas shift reaction: structure, function and biosynthesis of monofunctional [NiFe]-carbon monoxide dehydrogenases - Sustainable Energy & Fuels (RSC Publishing)

Water gas shift reaction for hydrogen production and carbon dioxide capture: A review - ScienceDirect

Question Video: Identifying the Chemical Formula of the Substance Produced When Carbon Dioxide Dissolves in Water | Nagwa

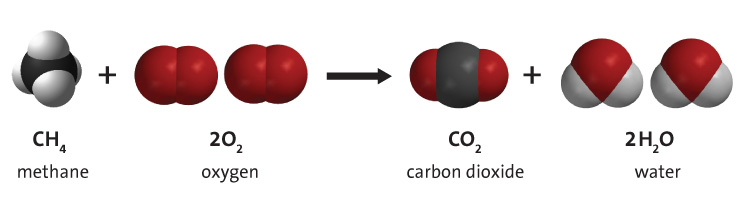
Multimedia: Carbon Dioxide Can Make a Solution Acidic | Chapter 6, Lesson 10 | Middle School Chemistry

Summary of the reactions between carbon dioxide (CO2) with water (H2O)... | Download Scientific Diagram
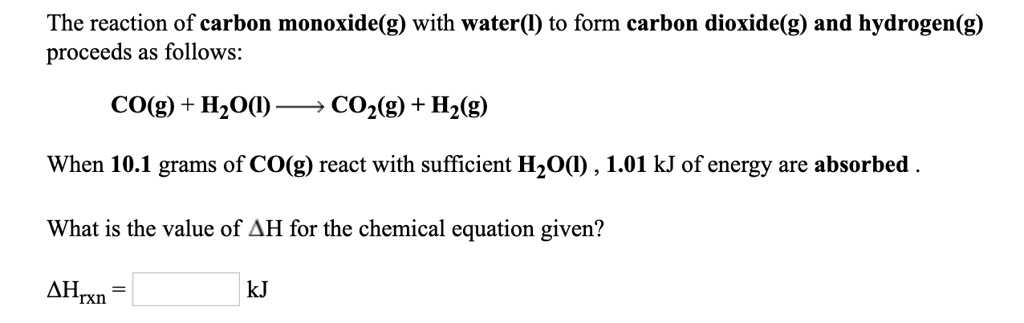
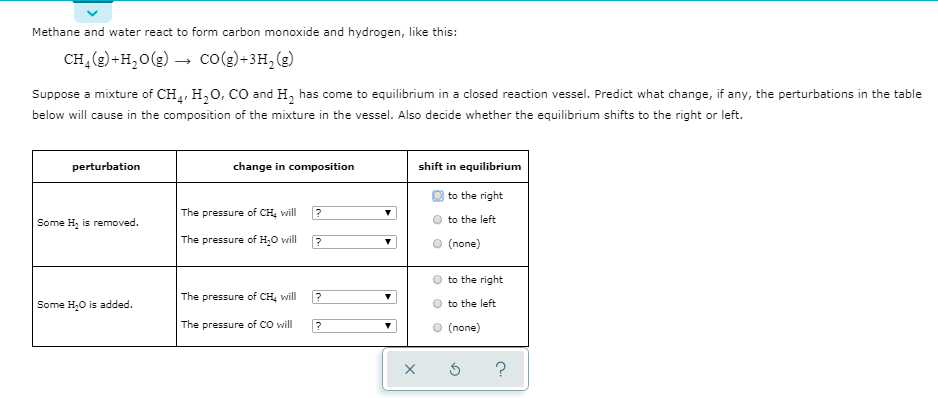
SOLVED: The reaction of carbon monoxide(g) with water() to form carbon dioxide(g) and hydrogen(g) proceeds as follows: CO(g) + HzO() COz(g) + Hz(g) When 10.1 grams of co(g) react with sufficient HzO()