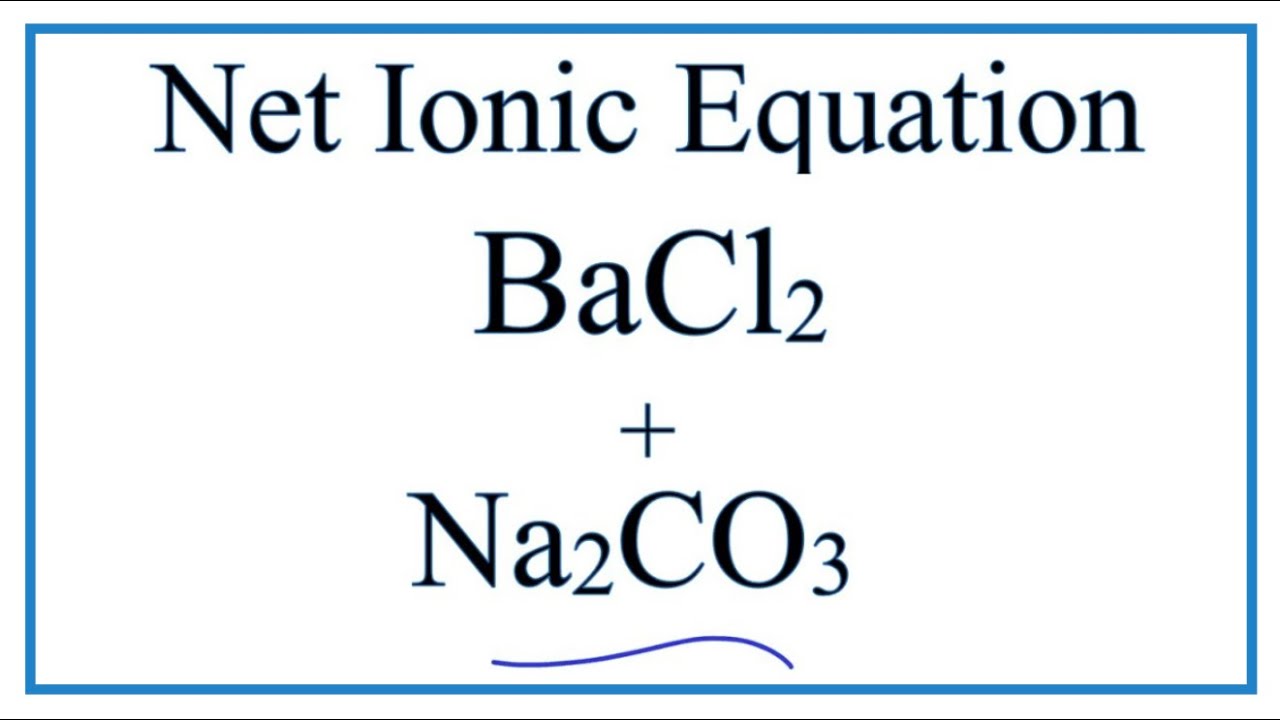To perform the reaction between sodium sulphate and barium chloride solutions and classify it - Lab Work

SOLVED: I@Lead(II) acetate and sodium carbonate (3Lead(II) acetate and sodium chloride - I4Lead(I) acetate and sodium sulfate SLead(II) acetate and sulfuric acid 'bLead(II) acetate and barium chloride I7Lead(II) acetate and hydrochloric acid

Net Ionic Equation for BaCl2 + Na2CO3 = BaCO3 + NaCl | Net Ionic Equation for BaCl2 + Na2CO3 = BaCO3 + NaCl This is an example of a double displacement reaction.
BaCl2 + Na2CO3 - barium chloride and sodium carbonate (double replacement precipitation reaction) - video Dailymotion

SOLVED: Experimental Section 1,Carbonate Ion 1. Pour of" sodium carbonate solution t0 lest tubeand add several drops of barium chloride. 1.2 Afier sedimentalion remove the solution over resulting precipitate and add several

Write a balanced chemical equation when barium chloride reacts with sodium sulphate to give insoluble barium sulphate and sodium chloride.
Sketch of the experimental setup. A barium chloride solution is pumped... | Download Scientific Diagram